Thermochemistry science, Types of systems & law of conservation of energy
Energy
Energy is very important in our life , where we can’t carry out different activities ( mental or muscular ) without the produced energy from burning sugar inside our bodies , we can’t cook our food without the heat energy produced from the burning of natural gas .
There are various forms of energy like Chemical energy , Heat energy , Light energy , Electrical energy and Kinetic energy , In spite of this classification of energy into different forms , There is a relationship between all of these forms , as the energy is converted from one form to another , this leads us to the law of conservation of energy .
Law of conservation of energy
The energy in any physical or chemical change can be neither created nor destroyed, but it is transformed from one form to another.
Thermochemistry science
All the chemical reactions and the physical changes are accompanied by changes in the energy , Thermochemistry is a branch of thermodynamics which concerned with these changes.
Thermochemistry is the science that concerns with the heat changes that accompanying the chemical changes ( the reactions ) and the physical changes , Thermodynamics is the science that deals with the study of energy and how it transfers .
The dissolving of ammonium nitrate salt in water represents a physical change , The combination of hydrogen and oxygen to form water represents a chemical change ( reaction ) .
Before studying of how we can calculate the heat changes accompanying the chemical and physical change , we have to know the following basic concepts: System and surrounding, First law of thermodynamics , Temperature and heat , Specific heat .
System and surrounding
System is the part of the universe chosen for study in which the physical or the chemical change occurs , Surrounding is the part outside the system that exchanges the energy with the system in the form of heat or work .
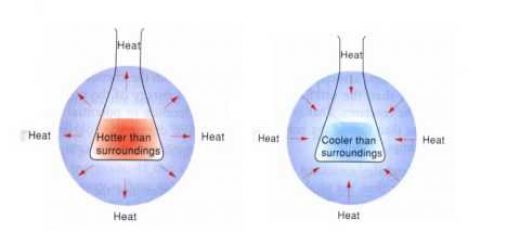
The chemical reactions are accompanied with changes in the energy ( releasing or absorbing ) , this energy exchange occurs between the reaction mixture and its surrounding , where :
- The system expresses the reactants and the products .
- The system boundary expresses the wall of the container ( a flask or a test tube ) in which the reaction occurs .
- The surrounding expresses the medium surrounding the reaction container ( the laboratory ) .
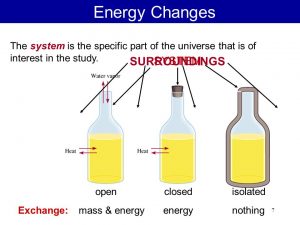
Types of systems
Systems are classified according to their ability to exchange the energy or matter with its surrounding into Open system , Closed system and Isolated system , Open system is the system that freely exchanges matter and energy with its surrounding , Closed system is the system that exchanges energy ( but not matter ) with its surrounding in the form of heat or work .
Isolated system is the system that exchanges neither energy nor matter with its surrounding , The system doesn’t interact with its surrounding, The medical thermometer is considered as a closed system because it allows the exchange of energy only with the surrounding in the form of heat.
First law of Thermodynamics
Any change in the system’s energy Δ E system is accompanied by a change in the surrounding energy Δ E surrounding by a similar value , but with an opposite sign to make the total energy remain constant .
Δ E system = − Δ E surrounding
First law of thermodynamics includes the study of the energy exchange between the isolated system and its surrounding .
First law of thermodynamics : The total energy of an isolated system is constant even the system is changed from one state to another .
Heat and temperature
You can read this article about Combustion ( bomb ) Calorimeter , Specific heat and calculation of the quantity of heat
Potential energy, Kinetic energy & Law of conservation of mechanical energy