Properties of Alkaline earth elements, What are common uses of Alkaline earth metals?, Why are they called “alkaline earth metals”?
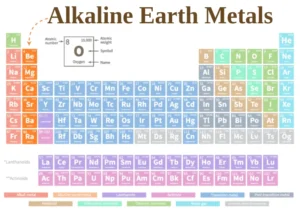
Alkaline earth elements are the metallic elements found in the second group of s-block after the group (1 A). They are located on the left side of the modern periodic table.
Alkaline earth elements
The alkaline earth metals are all shiny, silver-white, and they are good conductors of heat and electricity. The alkaline earth metals have similar properties. Their densities are higher than the densities of the alkali metals. All the alkaline earth metals sink in the water as they have higher densities than the water density.
The alkaline earth metals in group (2) have two electrons in their valence shells. This enables the metals to easily lose electrons, which increases their stability and allows them to form compounds. They are divalent elements as they have two electrons in their outermost energy levels.
The alkaline earth metals are reactive metals at standard temperature and pressure, but the chemical activity of these elements is less than that of the alkali metals activity. Therefore, they are not kept under the surface of the Kerosene or paraffin oil.
When we look at the modern periodic table, we will notice that the chemical activity of the alkaline earth metals increases as their atomic size increases, and the loss of the two valency electrons becomes easier.
Barium (Ba) is more active than Calcium (Ca), which is more active than magnesium (Mg). All alkaline earth metals react with hydrogen to create metallic hydrides, they all react with oxygen to produce metal oxides, and they all react with halogens to form ionic halides.
FAQ about properties of Alkaline Earth Metals
1. What are alkaline earth metals?
Alkaline earth metals are elements in Group 2A of the periodic table. They include: Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and Radium (Ra).
2. Why are they called “alkaline earth metals”?
They are called “Alkaline” because their oxides and hydroxides are basic (alkaline). “Earth metals” because they were historically found in mineral ores.
3. What is the electronic configuration of Group 2 elements?
They have two electrons in their outermost shell: ns² configuration, which makes them likely to lose two electrons and form +2 ions.
4. What is the common oxidation state of alkaline earth metals?
The most common oxidation state is +2, as they tend to lose two valence electrons.
5. How reactive are alkaline earth metals?
They are reactive metals, but less reactive than alkali metals (Group 1). Reactivity increases down the group (Be → Ba).
6. What are the physical properties of alkaline earth metals?
- Shiny, silvery metals.
- Harder and denser than alkali metals.
- Good conductors of heat and electricity.
- Higher melting and boiling points than Group 1 elements.
7. How do they react with water?
- Beryllium: does not react with water.
- Magnesium: reacts slowly with hot water.
- Calcium, Strontium, and Barium: react readily with cold water to form hydroxides and hydrogen gas.
8. What is the general reaction with oxygen?
They react with oxygen to form metal oxides (MO), which are basic in nature.
9. What are the properties of their oxides and hydroxides?
- Oxides (MO) are basic.
- Hydroxides (M(OH)₂) are alkaline.
- Basicity increases down the group.
10. What is the trend in atomic size down the group?
Atomic radius increases down the group due to the addition of electron shells.
11. How does ionization energy change in the group?
Ionization energy decreases down the group, making it easier for atoms to lose electrons.
12. What is the trend in the solubility of hydroxides?
Solubility of hydroxides increases down the group (Mg(OH)₂ is less soluble than Ba(OH)₂).
13. What is the trend in the solubility of sulfates?
Solubility of sulfates decreases down the group (BaSO₄ is nearly insoluble).
14. Why is beryllium different from other Group 2 elements?
Beryllium shows unusual behavior because:
- It has a small atomic size.
- High ionization energy.
- Forms covalent compounds rather than ionic.
15. What are the common uses of alkaline earth metals?
- Magnesium: light alloys, fireworks.
- Calcium: cement, bones and teeth.
- Barium: medical imaging (BaSO₄).
- Strontium: red fireworks.
16. Are alkaline earth metals found freely in nature?
No, they are not found in free form due to their reactivity. They exist as compounds in minerals.
You can follow science online on YouTube from this link: Science online
Elements of s-block, Properties of the first group elements 1A (Alkali metals) in the periodic table
The general properties of the alkali metals in the modern periodic table
Alkali metals compounds properties and uses (Sodium hydroxide & Sodium carbonate)
Radius property, Ionization potential, Electron affinity & Electronegativity
Chemical combination, Properties of Metals, Nonmetals & Noble (inert) gases