Matter construction and properties of molecules of matter, Why is studying molecules important?
The matter consists of very small structural units known as molecules. The molecule is the smallest part of matter that can exist freely, and it has the properties of matter.
Properties of the molecules of matter
The molecules of matter are in continuous motion, and there are intermolecular spaces and attractive forces among the molecules. The kinds of matter are elements, compounds, and mixtures.
The intermolecular spaces are the spaces that are found among the molecules of matter. The intermolecular force is the force that binds the molecules of matter together.
The molecules of one substance are alike in properties, but they differ from the molecules of the other substance. The molecules are composed of tiny structural units called atoms.
The differences in molecules of various substances arise from differences in the atoms involved in their structures and depend on the number and kinds of atoms and the way they combine.
Element molecule
The element is the simplest pure form of matter that can not be analyzed chemically into a simpler form. The element molecule is formed of similar atoms (one or more atoms) that combine together.
Monoatomic elements
There are elements molecules that are formed of only one atom (The Monoatomic), such as the solids as (Copper, Iron, Aluminum, Sulphur, Magnesium, and Carbon) and the liquids as mercury.
The noble gases are Helium, Neon, Argon, Krypton, Xenon, and Radon.
Diatomic elements
Some element molecules are formed of two atoms (Diatomic), such as
- The liquids are bromine.
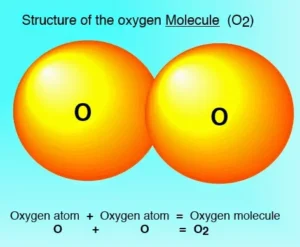
- The active gases are oxygen, Hydrogen, Chlorine, Nitrogen, and Fluorine.
| Oxygen molecule is an active gas, and the molecules of the active gases are diatomic. |
Compound molecule
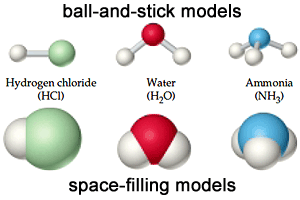
| The compound molecule is formed from different atoms. |
The compound molecule is formed of different atoms, and it is a substance that is formed from a combination of the atoms of two or more different elements with constant weight ratios.
Examples of compound molecules: Sodium chloride molecule (table salt), Water molecule, Ammonia molecule, Hydrogen chloride.
FAQ about Molecules of Matter
1. What is matter?
Matter is anything that has mass and occupies space. All objects around us, such as air, water, plants, and rocks, are forms of matter.
2. What is the basic construction of matter?
Matter is made up of tiny particles called atoms and molecules. These particles are extremely small and cannot be seen with the naked eye.
3. What is a molecule?
A molecule is the smallest unit of a substance that can exist independently while keeping the properties of that substance. It consists of two or more atoms chemically bonded together.
4. What are examples of molecules of matter?
Examples include:
- Water molecule (H₂O).
- Oxygen molecule (O₂).
- Carbon dioxide molecule (CO₂).
5. What are the main properties of molecules of matter?
Molecules of matter have several important properties:
- They are very small particles.
- There are spaces between them.
- They are in continuous motion.
- There are forces of attraction between them.
6. What is meant by the spaces between molecules?
The particles of matter are not tightly packed together, so there are spaces between them. These spaces differ depending on the state of matter (solid, liquid, or gas).
7. Why are molecules always in motion?
Molecules move continuously because they possess kinetic energy. Their movement becomes faster when the temperature increases.
8. What are the forces of attraction between molecules?
These are intermolecular forces that keep molecules close to each other. The strength of these forces determines whether matter exists as a solid, liquid, or gas.
9. How does the movement of molecules differ in the three states of matter?
- In solids, molecules vibrate in fixed positions.
- In liquids, molecules move more freely and slide past each other.
- In gases, molecules move very quickly and spread far apart.
10. Why is studying molecules important?
Studying molecules helps scientists understand the behavior of materials, chemical reactions, and physical changes, and it is important in fields such as chemistry, physics, and biology.
You can subscribe to Science Online on YouTube from this link: Science Online
Matter, Properties and Kinds of molecules, Melting process, and Vaporization process
Theories explaining the covalent bond, Octet rule & Overlapped orbitals concept