Importance of the ozone layer and its structure, What is the ozone hole?
The ozone layer is located at a height of 20 to 40 km above sea level, and it is located in the lower part of the stratosphere layer. As it is the first layer of the atmospheric envelope that contains a suitable amount of oxygen gas, and it also faces the ultraviolet radiations that emitted from the Sun.
Structure of the ozone layer
The ozone layer is composed of the ozone gas, which consists of three oxygen atoms.
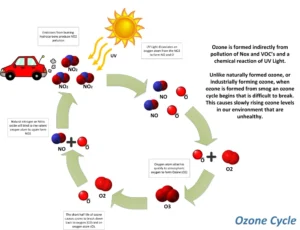
Formation of the ozone gas
| Each oxygen atom combines with oxygen, forming the ozone molecule |
The oxygen molecule absorbs ultraviolet radiation (UV), which causes the breakdown of the bond between the two oxygen atoms, giving two free oxygen atoms, and each oxygen atom combines with an oxygen molecule, forming the ozone molecule, which is composed of three oxygen atoms.
Thickness of the ozone layer
The value of the atmospheric pressure in the stratosphere layer is 0.001 of the normal atmospheric pressure at sea level, and the temperature is less than that at sea level. So, in these conditions, the zone gas forms a layer of a thickness of 20 km.
The English scientist “Dobson” postulated that the thickness of the ozone layer is compressed into 3 mm. Only if it is under the normal atmospheric pressure, and at the zero temperature (STP).
STP means standard temperature and normal atmospheric pressure.
So, the scientists took “Dobson” as a measuring unit of the degree of the ozone layer. The natural degree of the ozone is 300 Dobson units (100 Dobson units (DU) is defined as 1mm thickness).
FAQ about the Ozone Layer
1. What is the Ozone Layer?
The ozone layer is a region in the stratosphere of Earth’s atmosphere that contains a high concentration of ozone (O₃) molecules. It forms a protective shield that absorbs most of the harmful ultraviolet (UV) radiation from the Sun.
2. Where is the ozone layer located?
The ozone layer is mainly found in the stratosphere, about 15 to 35 kilometers above the Earth’s surface.
3. What is the structure of the ozone layer?
The ozone layer is composed of ozone molecules (O₃), which contain three oxygen atoms. These molecules are continuously created and destroyed through chemical reactions involving sunlight and oxygen in the stratosphere.
4. How is ozone formed in the atmosphere?
Ozone is formed when ultraviolet radiation from the Sun splits oxygen molecules (O₂) into single oxygen atoms. These atoms then combine with other oxygen molecules to form ozone (O₃).
5. Why is the ozone layer important?
The ozone layer is important because it protects life on Earth by absorbing most harmful ultraviolet (UV-B and UV-C) radiation, which can damage living organisms.
6. How does the ozone layer protect living organisms?
The ozone layer absorbs and filters dangerous UV rays before they reach the Earth’s surface, helping to prevent skin cancer, eye damage, and harm to plants and marine life.
7. What would happen if the ozone layer disappeared?
Without the ozone layer, Earth would receive extremely high levels of ultraviolet radiation, leading to severe health problems for humans, damage to ecosystems, and difficulty for many forms of life to survive.
8. What causes the depletion of the ozone layer?
The main cause of ozone depletion is human-made chemicals, such as chlorofluorocarbons (CFCs) found in old refrigerators, air conditioners, and aerosol sprays.
9. What is the ozone hole?
The ozone hole refers to a large area of severe ozone depletion, mainly observed over Antarctica during the spring season.
10. How can we protect the ozone layer?
The ozone layer can be protected by:
- Reducing the use of ozone-depleting chemicals.
- Using environment-friendly refrigeration systems.
- Supporting international agreements such as the Montreal Protocol.
11. Does the ozone layer help regulate Earth’s temperature?
Yes. By absorbing ultraviolet radiation, the ozone layer helps maintain the temperature balance of the stratosphere, which influences the overall climate system.
12. Is the ozone layer recovering?
Yes. Due to global efforts to reduce harmful chemicals, scientists have observed a slow recovery of the ozone layer, although full recovery may take several decades.
The ozone layer is a crucial part of Earth’s atmosphere that protects living organisms from harmful ultraviolet radiation. Its structure consists of ozone molecules formed through interactions between sunlight and oxygen, making it an essential shield for life on our planet.
You can subscribe to Science Online on YouTube from this link: Science Online
The erosion of the ozone layer and the protection from the ozone layer pollutants
The atmospheric pressure maps and the instruments of measuring the atmospheric pressure
Erosion of Ozone layer, Global warming phenomenon & Greenhouse effect