Importance of the water and its structure, Can life exist without water?, How can we conserve water?
There are no living organisms that can live without water, as it is the medium through which all the vital processes take place. The water in the oceans and the rivers is an important means of transportation between cities and countries.
The waterfalls are used to generate electricity in many countries. Consumption of water is very high in the world, where Asia is the largest continent in consuming water for agriculture, and North America is the largest continent in consuming water for personal uses.
Europe is the largest continent in water consumption in industry, and Africa is considered the agricultural continent, as the consumption of water is very high in the agricultural fields, since it has the best-cultivated lands.
Sources of the water
There are many sources of water, such as the water areas (such as the oceans, the rivers, the seas, ……………), the wells, the springs & the rain.
Structure of the water
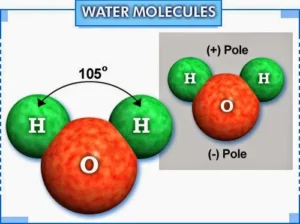
The water molecule is formed by the combination of one oxygen atom (O) with two hydrogen atoms (H) by two single covalent bonds, and the polar water molecules are linked together by “hydrogen bonds”.
| There are two single covalent bonds between two hydrogen atoms and one oxygen atom |
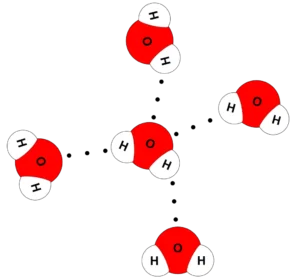
The hydrogen bond is a weak electrostatic attraction force that arises between the molecules of polar compounds, such as water molecules. The hydrogen bond is responsible for the abnormal properties of water, and it is weaker than the covalent bond.
| Polar water molecules are linked together by hydrogen bonds |
Water in the human body
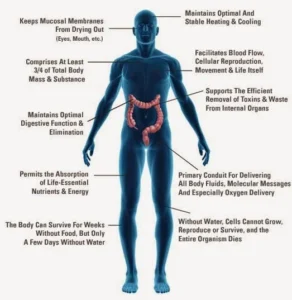
The human body is the most abundant substance in the living tissues, and makes up 70 percent or more of most forms of life.
| Without the water, the cells can’t grow, reproduce, or survive |
The water in the biological tissue can be classified into two types. The first type is called the bound water, and the second type is called the bulk water.
FAQ about the Water
Water is one of the most important substances on Earth. It is essential for life, supports ecosystems, and plays a key role in many biological and chemical processes. Below are frequently asked questions about the importance of water and its molecular structure.
1. What is water?
Water is a chemical substance composed of two hydrogen atoms and one oxygen atom. Its chemical formula is H₂O. It exists in three states: solid (ice), liquid (water), and gas (water vapor).
2. What is the structure of a water molecule?
A water molecule consists of:
- Two hydrogen atoms.
- One oxygen atom.
The hydrogen atoms are bonded to the oxygen atom by covalent bonds, forming a bent or V-shaped structure. This shape gives water unique physical and chemical properties.
3. Why is water important for life?
Water is essential for all living organisms because it:
- Makes up a large portion of cells and body fluids.
- Helps transport nutrients and oxygen.
- Removes waste products from the body.
- Regulates body temperature.
- Supports chemical reactions inside cells.
4. What percentage of the human body is water?
The human body is composed of about 60–70% water, depending on age and body composition.
5. Why is water called the universal solvent?
Water is called the universal solvent because it can dissolve many substances, such as salts, sugars, and gases. This property allows nutrients and minerals to be transported in living organisms and in nature.
6. What are hydrogen bonds in water?
Hydrogen bonds are weak attractions between water molecules. They form when the hydrogen atom of one water molecule is attracted to the oxygen atom of another molecule. These bonds are responsible for many special properties of water.
7. What are the main properties of water?
Important properties of water include:
- High heat capacity.
- High surface tension.
- Ability to dissolve many substances.
- Density changes when freezing.
- Strong cohesion and adhesion forces.
These properties make water essential for life and environmental processes.
8. Why is water important for plants?
Water helps plants by:
- Transporting nutrients from the soil.
- Supporting photosynthesis.
- Maintaining cell structure and plant growth.
- Cooling plants through transpiration.
9. What role does water play in the environment?
Water plays a vital role in:
- The water cycle (evaporation, condensation, precipitation).
- Climate regulation.
- Supporting aquatic ecosystems.
- Maintaining soil moisture for agriculture.
10. Can life exist without water?
Most known forms of life cannot survive without water. It is essential for metabolism, nutrient transport, and cellular processes.
11. Why does ice float on water?
Ice floats because it is less dense than liquid water. When water freezes, hydrogen bonds form a structure that spreads the molecules apart, making ice lighter than liquid water.
12. How can we conserve water?
Water conservation can be achieved by:
- Fixing leaks.
- Reducing water waste.
- Using efficient irrigation systems.
- Recycling water when possible.
- Protecting natural water sources.
You can subscribe to Science Online on YouTube from this link: Science Online
Water properties, structure, pollutants, & protection of water from pollution
The physical and chemical properties of the water
The water pollutants and the protection of the water from pollution