General properties of the first transition elements in the modern periodic table
Most of the transition elements are attracted to the external magnetic fields because the spin of unpaired electrons in the sublevel (d) produces a magnetic field that makes the atom attracted to the external magnetic field, The density of the elements of the first transition series increases by increasing the atomic number because the atomic number increases & the atomic mass increases while the size of the atom decreases slightly.
First transition series
The first transition series is a group of chemical elements that occupy the 4th period of the periodic table. They are also known as the 3rd transition series because their outermost electrons are filling the 3rd subshell. These elements are characterized by their transitional properties between the s-block and p-block elements. They often exhibit multiple oxidation states, form colored compounds, and have catalytic properties.
The elements in the first transition series are:
- Scandium (Sc)
- Titanium (Ti)
- Vanadium (V)
- Chromium (Cr)
- Manganese (Mn)
- Iron (Fe)
- Cobalt (Co)
- Nickel (Ni)
- Copper (Cu)
- Zinc (Zn)
Atomic Mass
The atomic mass of the first transition elements increases gradually with increasing their atomic number but nickel is abnormal because it has five stable isotopes with average mass 58.7U, There is a little change in atomic radii as we move through first transition series, Atomic radius of the transition elements is relatively constant from (Cr) to (Cu), this due to two opposite factors:
The first factor causes decreasing in the atomic radius with increasing the atomic number, where the effective nuclear charge for these elements increases and also the number of electrons in the atom increases from (Sc) to (Cr), so the nuclear attraction to electrons increases which cause decrease the atomic radius.
The second factor causes an increase to the atomic radius, The increase in the number of the electrons in 3d sublevel will increase the repulsion force between them, As a result of these two opposite factors, the atomic radii of these elements are relatively constant which explains using them in making alloys.
There is a gradual decrease in the atomic size from 21Sc till 29Cu, (relatively constant from Cr to Cu), Transition elements have small atomic size and variation in atomic size is small, it is difficult to oxidize the elements due to the slight decrease in radius & the increase of the nuclear attraction force to the valency electrons.
Due to the atomic radius is relatively constant, these elements are used in alloys industry, when the atomic radii are the same, the properties are similar, this explains the similarity in properties in Iron triad (Fe, Co, Ni), The density of iron (7.87 gm/cm³) is less than that of cobalt (8.8 gm/cm³) because the atomic mass of iron is less than that of cobalt while the difference in their radii is slight.
The gradual decrease in atomic radius across the elements of the first transition series from left to right is not large because the repulsion of the electrons added in the orbital compensates the decreasing in the radius due to increasing of the nuclear attraction to electrons as the effective nuclear charge increases.
The metallic property
The metallic property appears in transition elements due to:
- All transition elements are solid characterized by metallic luster, and good conductor of heat and electricity, They have high melting & boiling points due to the strong metallic bond which is formed due to the sharing of both 4S and 3d electrons in the formation of this bond.
- Most of the transition elements have high density, and density increases by increasing the atomic mass, where the atomic size (atomic volume) is relatively constant.
- There is variation in the activity of these elements as copper has limited chemical activity, and some are moderate like iron which goes rusty if it is exposed to air, and some are highly active like scandium which replaces Hydrogen in water strongly.
The density of transition elements increases from left to right, where the density of iron (7.87 gm/cm³) is less than that of cobalt (8.8 gm/cm³) because density = Atomic mass/ Atomic volume, and the atomic mass of iron is less than that of cobalt and atomic volume of (Fe & Co) is nearly equal.
Boiling and melting points
All transition elements have high melting points and boiling points due to the strong binding between their atoms where the electrons of (4S) & (3d) form strong metallic bonds, Transition elements are harder than representative elements due to increasing the free valency electrons (4S2 + 3d unpaired electrons), so the strength of metallic bond increases, Number of valency electrons ∝ strength of the metallic bond ∝ melting point and boiling point.
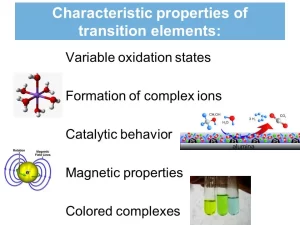
Cu and Zn are relatively soft and of lower boiling points as their metallic bonds are weaker since they involve the participation of 4S electrons only, The transition elements have some properties which differentiate them from the representative elements, these properties are magnetic properties, the coloured compounds, catalytic activity of the elements & the compounds.
Magnetic properties
There are two different magnetic properties which are paramagnetic property and diamagnetic property, Transition elements are classified into paramagnetic substances and diamagnetic substances, most of the compounds of the transition elements are paramagnetic:
The paramagnetic property appears in the ions or atoms or molecules, that have unpaired electrons in their orbitals (↑) which form a magnetic field due to its spinning, attracting an external magnetic field, The paramagnetic substance is the substance that is attracted to the external magnetic field due to the presence of unpaired electrons in the d-sublevel which spin in one direction forming a magnetic field.
The magnetic moment of the paramagnetic substances increases with the increase in the number of unpaired electrons, most of the compounds of the transition elements are paramagnetic because of the spinning movements of the electrons.
Maximum number of unpaired electrons = 5
Magnetic moment = Number of unpaired electrons in d sublevel
For unpaired electrons, the spin will be in one direction (↑) which produces a magnetic field and forms a paramagnetic substance while for paired electrons, the spin will be in opposite directions (↑↓) and there is no magnetic field and forms a diamagnetic substance.
The diamagnetic property appears in substances in which all orbitals are paired with electrons (↑↓), so, their magnetic moment is equal to zero due to the opposite spin motion of every paired electrons, A Diamagnetic substance is a substance that repels with the external magnetic field due to the presence of paired electrons in the d sublevel which spins in opposite direction, so, the magnetic moment = zero.
The magnetic moments of the substance can be measured by the number of unpaired electrons, The measure of magnetic moments of the substance indicates the number of unpaired electrons and the electronic configuration of the metal ion.
Measuring the magnetic moment helps in determination the electronic configuration because when the magnetic moment = zero, this means that all electrons are coupled when it has a certain value this means the presence of a number of unpaired electrons and the magnetic moment is directly proportional to the number of unpaired electrons.
FeCl3 is attracted to the magnet more than TiCl3 because Fe+3 has 5 unpaired electrons, while Ti+3 has only one electron, accordingly, the magnetic moment of Fe+3 is more than Ti+3, so FeCl3 is attracted to the magnet more than TiCl3.
Catalytic activity
Transition elements are considered as an ideal catalyst where finely divided nickel is used as a catalyst in the hydrogenation process of oil, divided iron is used as a catalyst in the preparation of ammonia gas NH3 by the Haber-Bosh method.
N2 + 3H2 → 2 NH3
Vanadium pentoxide V2O2 is used as a catalyst during the preparation of sulphuric acid by the contact method.
2 SO2 + O2 → 2 SO3
SO3 + H2O→ H2SO4
The catalytic activity of the metals of the first transition series refers to the presence of the 4S and 3d electrons which can be used in the formation of bonds between the atoms of the surface of metal and the reacting molecules leading to the increase of concentration of these molecules on the surface of the catalyst weaken the bond in the reactant molecules and so decrease the activation energy which help to increase speed of reaction.
Elements of the first transition series are good catalysts, finely divided nickel is used as a catalyst for hydrogenation of oil due to the presence of the 4S and 3d electrons which form bonds between the reacting molecules and the surface of the catalyst which leads to:
- Increasing the concentration of the reactants at the surface of the catalyst.
- The rate of collision between the reacting molecules increases.
- The bonds in the molecules of the reactants become weak.
- The activation energy decreases and the speed of the reaction increases.
Coloured ions
Most of the compounds of transition elements and their aqueous solution are coloured, The colour is formed due to the absorption of some photons in the area of visible light, Eye sees only the remained colour (reflected) which is a mixture of some colours.
The light is a form of energy and each compound of transition elements especially which has unpaired electrons in the d sublevel when exposed to light their electrons are excited by a certain colour of white light, (white light consists of 7 colours), so, the substance absorbs it and does not absorb others (complementary), which we see substance by it.
- If the substance absorbs no light colour, it appears white (colourless), it takes place in case of d0 and d10, in case of ions of representative elements, where they have unpaired electrons in the sublevel S and P where the energy of the visible light can not excite them to higher energy levels, so they are colourless.
- If the substance absorbs all visible light colour, it appears black to the eye.
- If the substance absorbs a certain colour, its colour appears by the complementary colour.
The hydrated ions of Sc+3( 3d0), Zn+2 ( 3d10) are colourless also nontransition elements because they contain orbitals of d empty ( d0) or completely filled ( d10), then we can conclude that the colour in the transition elements ions is due to partially filling of the orbitals of d sublevel (1→9) electrons due to the presence of the unpaired electrons in d sublevel.
Complementary colours are the net of colours which are not absorbed by the substance, They are the colours seen, Example: Chromium (III) compounds absorb red colour, so it appears green, There are some compounds although the (d) sublevel is d0 or d10 , they are coloured such as KMnO4 is violet although Mn+7.
Cobalt compounds appear with blue colour because the energy required exciting the unpaired electrons in the 3d-sublevel is equal to the energy of the orange colour, so, it appears with the complementary colour which is the blue colour.
Cu+1 is colourless because the sublevel d is completed d10, the electrons of the sublevel 3d don’t absorb any colour of the white light and reflect it, while Cu+2 is blue because the sublevel d is not completed d9 , The sublevel is not completed filled, so the unpaired electrons absorb one of the seven colours and the complementary colour is seen.
Solutions of cobalt salts are coloured because 3d7 sublevel is partially filled which means there are many unpaired electrons in the d-sublevel, so it absorbs a colour from the white light and complementary colour is seen, whereas solutions of zinc salts are colourless because 3d10 sublevel is completely filled which means, there are no unpaired electrons in the d-sublevel, so it doesn’t absorb any colour.
First transition elements properties, electronic configuration & oxidation states
Extraction of Iron from its ores, Iron dressing, Reduction & production