Physical and chemical properties of Esters, Esterification reaction and preparation of ethyl acetate ester
Esters are the products of the combination of carboxylic acids with alcohols, Esters are widely spread in nature, They are present in all plant and animal organisms, Most of the esters are characterized by a pleasant smell which is responsible for the odour and flavour of fruits and flowers.
Esters
Many organic esters have been prepared for the commercial production of perfumes and flavours, They are used either alone or mixed with natural compounds, The odour of the ester decreases with the increase of the molecular weight of the alcohol and acid used for their formation.
The nature of the ester changes from a liquid with a pleasant odour to a waxy solid which is nearly odourless, waxes such as bee wax are high molecular weight esters, and Fats and oils are esters derived from glycerol which is trihydric alcohol and high fatty acids.
The name of an ester is derived from the name of the acid radical and the name of the alkyl group of the alcohol such as Methyl formate HCOOCH3, Ethyl acetate CH3COOCH2CH3, and Ethyl benzoate C6H5COOC2H5.
Preparation of ethyl acetate ester
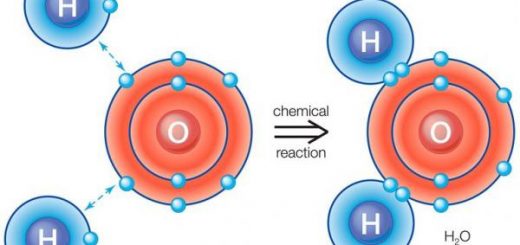
The direct method for the preparation of esters is the reaction between the carboxylic acid and the alcohol, For example, the ethyl acetate ester is obtained by the reaction of acetic acid and ethyl alcohol.
CH3COOH + C2H5OH ⇔CH3COOC2H5 + H2O
This reaction is reversible where the formed ester is hydrolyzed to the acid and alcohol, It is preferred to use a dehydrating agent such as concentrated sulphuric acid or hydrogen chloride gas to get rid of the formed water.
The concentrated sulphuric acid is used in preparing ethyl acetate ester to get rid of water to be sure that the reaction is shifted to the forward direction (direction of ester formation).
C6H5COOH + C2H5OH ⇔C6H5COOC2H5 + H2O
In the direct method to prepare Ethyl Benzoate Ester, Dry HCl is used for dehydration, not concentrated H2SO4 to avoid the sulphonation of benzoic acid.
Physical properties
Most of the esters are liquids with much lower boiling points than those of the acids or alcohols of nearly equal weight, this is due to the absence of the polar hydroxyl group which is found in alcohols and acids and leads to the association of the alcohol and carboxylic acid molecules with hydrogen bonds, Esters have a neutral effect on litmus.
The boiling point of the ester is less than the boiling point of the acid and alcohol forming it due to the absence of polar hydroxyl group (presents in alcohols and acids) which can form hydrogen bonds between molecules and water.
The solubility degree of ester in water is less than that of the corresponding acid due to the absence of polar hydroxyl group (presents in alcohols and acids) which can form hydrogen bonds between molecules and water.
Chemical properties
Hydrolysis of esters: Alcohol and acid are produced from the hydrolysis of ester, this reaction reverses to ester formation, Hydrolysis may take place by the use of dilute mineral acids as a catalyst and is called acid hydrolysis, Dilute mineral acid (H+) is used to prevent the reversible reaction.
CH3COOC2H5 + H2O → CH3COOH + C2H5OH
Hydrolysis of esters may also be carried out by heating with aqueous alkalies, to produce the alcohol and the salt of the acid, this is called alkaline-hydrolysis or saponification, (since soap is the sodium salt of high carboxylic acids), NaOH is added to react with the produced acid converting it to salt and prevent the reversible reaction.
CH3COOC2H5 + NaOH → CH3COONa + C2H5OH
C6H5COOC2H5 + NaOH → C6H5COONa + C2H5OH
Hydrolysis of esters is the reaction of the ester with water (in an acidic medium) to form acid and alcohol, saponification is the heating of ester with aqueous alkalis to produce the alcohol and the salt of the acid, Soap is the sodium salt of high fatty carboxylic acids.
The hydrolysis product of ester depends on the reaction medium because in an acidic medium, acid and alcohol are formed while in an alkali medium, salt of organic acid and alcohol are formed, Ammonolysis is the reaction of esters with ammonia to produce acid amide and the alcohol.
CH3COOC2H5 + NH3 → CH3CONH2 + C2H5OH
C6H5COOC2H5 + NH3 → C6H5CONH2 + C2H5OH
The esterification reaction is not a neutralization reaction as it is a reversible reaction “incomplete”, It needs a dehydrating agent, It is a slow reaction because it takes place between molecules, water is formed due to the combination of molecules, H from alcohol and OH from acid.
Acid + alcohol ↔ ester + H2O
The neutralization reaction is an irreversible reaction “complete”, It is a fast reaction because it takes place between ions, water is formed due to the combination of ions H+ from acid and OH− from alkali.
Acid + base → salt + H2O
Organic acids in our life, Aromatic carboxylic acids, Benzoic acid preparation & properties