Organic acids in our life, Aromatic carboxylic acids, Benzoic acid preparation and properties
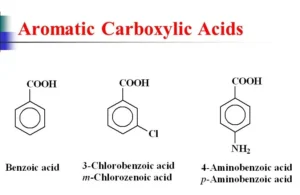
Aromatic carboxylic acids are compounds containing one or more carboxylic groups attached to the benzene ring. Benzoic acid is an example of a monocarboxylic (monobasic acid). Phthalic acid is an example of a dicarboxylic (dibasic) aromatic acid. Aromatic acids are stronger than aliphatic acids due to the acidity of the benzene ring.
Preparation of benzoic acid
Benzoic acid can be prepared by the oxidation of toluene or benzaldehyde by the proper oxidizing agent. It is prepared commercially by the oxidation of toluene in atmospheric air at 400°C and in the presence of vanadium pentoxide V2O5.
2C7H8 + 3O2 → 2 C7H6O2 + 2 H2O
Aromatic acids are generally stronger, less soluble in water, and less volatile than aliphatic acids. The reaction of the carboxylic group resembles the aliphatic acids, this can be by the formation of salts with metals, their hydroxides or carbonates, and the formation of esters with alcohols.
Physical properties of Benzoic acid
- Benzoic acid is less soluble in water than acetic acid because the molecular weight of benzoic acid is greater than that of acetic acid, and as the molecular mass increases, the solubility decreases.
- Benzoic acid is less volatile; it has a higher boiling point than acetic acid because the molecular mass of benzoic acid is greater than that of acetic acid.
- Benzoic acid is stronger in acidity than acetic acid.
Carbolic acid < Acetic acid < Benzoic acid < Salicylic acid < Phthalic acid
The organic acids in our life
Formic acid (HCOOH) is the acid that is secreted by ants to defend themselves. It is used in the manufacture of dyes, insecticides, perfumes, drugs, and plastics.
Acetic acid CH3COOH (pure 100%) isa colourless liquid, corrosive to skin, and it has a pungent odour. It freezes at 16°C forming solid substances as ice, therefore it is called glacial acetic acid (100% concentration), and it is used in esterification reactions to prevent the reversible reactions.
Acetic acid of concentration 4% is used as vinegar in homes. It is considered as a starting material for the synthesis of many organic products such as synthetic silk, dyes, insecticides and food additives.
Benzoic acid (C6H5COOH) is sparingly soluble in water, It is converted to its sodium or potassium salts to become soluble in water and to be easily absorbed in the human body, sodium benzoate of concentration of 0.1% is used as a preservating substance for foods because it prevents the growth of fungi on foods.
Citric acid is found in fruits, its percentage is (5–7%) in lemon and 1% in orange. It prevents the growth of bacteria in foods because it decreases their (PH), It has many industrial uses, and it is added to fruits to retain their colour and taste.
Lactic acid (C3H6O3) is found in milk as a result of the effect of enzymes secreted by certain kind of bacteria on milk sugar (lactose), It is generated in the human body as a result of hard effort, and causes a constriction in muscles, The football players are infected by a constriction in muscles during playing due to lactic acid which is generated in the human body as a result of hard effort and causes a constriction in muscles.
Ascorbic acid “vitamin (C)” is found in acidic substances like fruits, vegetables, and green pepper, it decomposes by heat and the effect of air, Lack of vitamin C leads to deterioration of some biological functions in the human body and infection with (Escarpot) disease, from its symptoms, bleeding of gum and swelling of the joints.
The human body needs vitamin C in small quantities. The fruits and some vegetables are eaten fresh without cooking because ascorbic acid (vitamin C) found in them is hydrolyzed by heat.
Salicylic acid is used in the manufacture of cosmetics specific to skin because it makes it softer, flexible and protect it against sun rays, and It is used in the elimination of skin warts and acne, It is also used in preparation of Aspirin and Marookh, It was used in the treatment of cold diseases and headache since 1829, before using Aspirin, but it caused bleeding of stomach.
Amino acids are known as organic acid derivatives, The amino derivatives of organic acids contain a carboxylic group (–COOH) and amino group (–NH2), the simplest number of amino acids is glycine which is known also amino acetic acid, It is formed by replacing one hydrogen atom in the alkyl group of the acid molecule by an amino group (–NH2).
There are many amino acids in nature, but 20 of them are found in natural proteins, The amino acids which form proteins are of the (α) amino type, the amino group is attached to (α) carbon atom which is directly attached to the carboxylic group, Glycine is the basic unit in the manufacture of protein, Glycine is called alpha (α) amino acetic acid because the amino group is attached to the (α) carbon atom which is directly attached to the carboxylic group.
Proteins are considered to be polymers of amino acids. Insulin, for example, is a protein molecule of molecular mass 6000; it consists of 51 molecules of 16 amino acids. The molecular mass of some protein molecules may reach one million more in the same complicated enzymes.
FAQ About Organic Acids in Our Life, Aromatic Carboxylic Acids & Benzoic Acid
What are organic acids?
Organic acids are carbon-containing compounds that have one or more –COOH (carboxyl) functional groups. They are weak acids and widely found in nature.
Where do we find organic acids in daily life?
Organic acids are present in:
- Fruits (citric acid in lemons).
- Vinegar (acetic acid).
- Yogurt (lactic acid).
- Medicines (acetylsalicylic acid – aspirin).
- Food preservatives (benzoic acid).
What are the uses of organic acids?
- Food preservation.
- Flavoring agents.
- Pharmaceutical production.
- Cleaning agents.
- Cosmetic products.
Why are organic acids important?
They:
- Regulate metabolism in living organisms.
- Help in digestion.
- Prevent bacterial growth.
- Are essential in industrial chemical production.
What are aromatic carboxylic acids?
They are organic acids in which the –COOH group is directly attached to an aromatic ring (like benzene).
What is the general formula of aromatic carboxylic acids?
Ar–COOH, where “Ar” represents an aromatic ring.
Give examples of aromatic carboxylic acids.
- Benzoic acid.
- Salicylic acid.
- Phthalic acid.
What are their properties?
- Weak acids.
- Undergo electrophilic substitution reactions.
- More acidic than phenols.
- Slightly soluble in cold water.
What are their uses?
- Food preservatives.
- Production of dyes and perfumes.
- Pharmaceutical manufacturing.
What is benzoic acid?
Benzoic acid (C₆H₅COOH) is the simplest aromatic carboxylic acid.
How is benzoic acid prepared?
- Oxidation of toluene: Toluene is oxidized using potassium permanganate (KMnO₄). The methyl group converts into a carboxyl group.
- From benzotrichloride: Hydrolysis produces benzoic acid.
- From Grignard reagent: Reaction of phenyl magnesium bromide with CO₂ followed by acidification.
What are the physical properties of benzoic acid?
- White crystalline solid.
- Slightly soluble in cold water.
- More soluble in hot water.
- Melting point ≈ 122°C.
What are the chemical properties of benzoic acid?
- Reacts with bases to form benzoates.
- Forms esters with alcohols.
- Undergoes substitution reactions in the benzene ring.
- Shows acidic behavior.
What are the uses of benzoic acid?
- Food preservative (E210).
- Antifungal agent.
- Preparation of dyes and perfumes.
- Pharmaceutical production.
You can follow science online on YouTube from this link: Science online
Acetic acid, Aliphatic carboxylic acids types, nomenclature, preparation and general properties
Types of compounds, Properties of Acids, Bases (alkalis), Oxides and Salts