Thermal emission, photoelectric effect, Cathode ray tube and Photoelectric cell
Any metal contains positive ions & free electrons which can move around inside the metal but can not leave it due to the attractive forces of the surface which may be represented by a surface potential barrier, Some of these electrons can escape if given enough energy in the form of heat and overcome the forces of attraction at the surface.
This is the idea behind the cathode ray (CRT) which is used in TV, computer monitors and photoelectric cells, the surface potential barrier is the attractive force that attracts the electrons from inside & prevents them from liberating from the metal surface.
Cathode ray tube
Cathode ray tube is used in television and computer monitors, It depends on the emission of electrons from the surface of the metal at heating (Thermoelectric effect).
Structure and working method
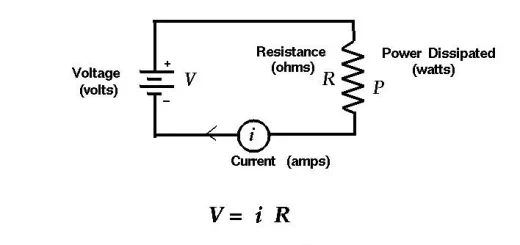
The tube consists of a metal surface called the cathode, The cathode is heated by a filament, and electrons are emitted from the electron gun (E-gun), due to heat, some electrons may overcome the forces of attraction at the surface (surface potential barrier).
These electrons are liberated from the metal and then picked up by the screen, which is connected to a positive pole called an anode, thus causing current in the external circuit, When the electrons hit the screen, they emit light which varies in intensity from point to point on the fluorescent screen, depending on the intensity of the electrical signal transmitted.
The energy of electrons (K E) is determined from the relation: K E = ½ me v² = e V
The energy (Joule) = Energy in electron Volt × Charge of the electron
Where: [(me) is the electron mass, (V) is the average velocity of the electron, (e) is the electron charge, (V) is the potential difference between cathode and anode].
The intensity of the electron beam can be controlled by using a grid in the way of the electrons as well as using magnetic and electric fields (plates X, Y) to direct the bundle of electrons to sweep the screen point by point generating the picture, so, called raster until the frame is completed.
Photoelectric cell
It is used to convert light energy into electric energy as in the calculators, opening and closing the doors, and some instructions, It depends on the emission of electrons due to falling of light on a metallic surface (photoelectric effect).
Photoelectric cell structure
- The photoelectric cell consists of a metallic surface which is called the cathode, This wire is called an anode to do not hide the light falls on the cathode.
- When light falls on the metallic surface, some electrons take enough energy to escape.
- These electrons are picked by anode which causes the current in the external circuit.
Observation:
- The emission of the electrons (called photoelectrons) depends on the frequency of the incident light not the intensity of the light, Such that these electrons will not be emitted until the frequency of the incident light becomes greater than or equal to a certain value which is called the critical value (νc) however is the intensity.
- If the frequency of the incident light wave is equal or greater than νc, so, the intensity of the photoelectric current (the number of electrons) increases by increasing the intensity of the incident light to increase the number of incident photons on unit area of a surface, so, the number of electrons liberate from the surface increases.
- The velocity and the kinetic energy of the emitted electrons depend on the frequency of the incident light not its intensity.
- The emission of the electrons occurs instantly as long as ν > νc, The electrons do not need time to collect energy even if the light intensity is low.
Classical theory failed to explain the photoelectric phenomenon because based on classical postulates
- The emission of electrons (photoelectrons) depends on the intensity of the incident wave regardless of its frequency.
- The kinetic energy (or velocity) of the emitted electrons increases with increasing the intensity of the incident radiation.
- Even in the case of low light intensity, giving sufficient time should give some electrons enough energy to be liberated, regardless of the frequency of the incident light.
Einstein explanation
Einstein put an interpretation for all of this, which led him to the Nobel prize in Physics in 1921, He explained the photoelectric phenomenon as follows: A certain amount of energy is needed to liberate the surface electrons called the work function (Ew).
For every metal there is a work function (Ew), if the falling photon has the energy (h ν) which is less than the work function (Ew) of the metallic surface, the electron would not be emitted at all no matter how intense the light might be and the emission is instantaneous.
If the falling photon has the energy (h ν) which is equal to the work function (Ew) of the metallic surface, the electron will be barely set free without any energy in this case, the frequency of the photon is equal to the critical frequency (νc).
Ew = h νc = h c / λc
If the falling photon has the energy (hν) which is greater than the work function (Ew) of the metallic surface, the electron will set free and the energy difference (h ν − Ew) is carried by the electrons as kinetic energy and it moves faster.
The photoelectric electrons are the emitted electrons from a metallic surface due to light falling with suitable frequency, Work function (Ew) is the minimum energy needed to free an electron from a metallic surface without gaining kinetic energy.
When the work function of zinc metal = 6.89 × 10−19 J, It means that the minimum energy needed to liberate an electron from the zinc metal surface without gaining kinetic energy = 6.89 × 10−19 J.
The critical frequency of a metal (νc) is the minimum frequency of light falling on a metallic surface to liberate the electron without gaining kinetic energy.
When the critical frequency of a surface = 4.8 × 1014 Hz, It means that the minimum frequency of light falling on the metallic surface to liberate the electron without gaining kinetic energy = 4.8 × 1014 Hz.
The photoelectric phenomenon is the emission of electrons from a metallic surface due to the falling of light with suitable frequency.
The factors affecting the work function (Ew) of a metallic surface:
- The type of material only.
- It does not depend on the light intensity, exposure time or potential difference between the anode and the cathode.
The relation between photocurrent and light intensity
If the frequency of the incident photon is less than the critical frequency of a surface, the photocurrent will not be emitted even if the light intensity or the time of falling of light increased, ν < νc.
If the photon frequency of incident light is greater than the critical frequency of a surface, the photocurrent intensity increases by increasing the light intensity (increasing number of photons), ν > νc.
The relation between the kinetic energy of the emitted electrons from a metal surface and the frequency of the incident light:
The energy of the incident photon = The work function of a metallic surface + The kinetic energy of the emitted electrons.
E = Ew + K E
h ν = h νc + ½ mv², This is Einstein’s equation for the photoelectric phenomenon.
The electron that is more bonded needs more energy than the work function to escape while the electron at the surface, needs energy equal to the work function to be liberated.
Quantum physics, Blackbody Radiation & importance of studying emitted ray from different bodies
Compton effect, Photon properties, Electron microscope and Optical microscope