Boiling point and separation of the petroleum oil, Fractional distillation of crude oil steps
The boiling point is the temperature at which a matter begins to change from a liquid state to a gaseous state. The change of matter from the liquid state to the gaseous state is known as boiling, and the temperature at which the matter begins to boil is called the boiling point.
Boiling point
The boiling point of the water is 100° Celsius, and this means that the water begins to boil and change into water vapor (gas).
The pressure affects the boiling points, and the boiling points of the liquids vary widely at normal atmospheric pressure. Because of this variation in the boiling points, two or more liquids can often be separated by a process called fractional distillation.
At the boiling point, the atoms or the molecules of the liquid gain enough energy to change the liquid into water vapor, and then the boiling rapidly evaporates.
Life application of the boiling process
The separation of petroleum oil depends on the difference between them in their boiling points. The separation of the components of the oil can be done by heating the crude oil and then separating each substance at its boiling point.
The boiling point is the temperature at which the vapour pressure of the substance is equal to the atmospheric pressure, The boiling point depends on the pressure, whereas the pressure increases, the boiling point increases.
The pressure pans are used for fast cooking as they raise the pressure so, the boiling point increases and the food is cooked faster.
Separation of the petroleum oil
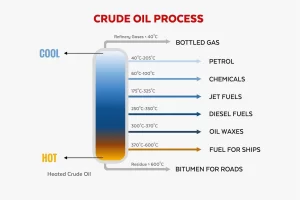
The separation of petroleum oil is also known as crude oil refining. It is a process that involves breaking down crude oil into its useful components. This process is primarily carried out through fractional distillation and other refining techniques to produce fuels and other products.
1. Desalting
Crude oil is often contaminated with water, salts, and sediments. The first step in refining is desalting, where these impurities are removed by washing the crude oil with water. This helps prevent corrosion in the refining equipment.
2. Fractional Distillation
This is the primary method used to separate crude oil into different fractions based on boiling points. Crude oil is heated to about 350-400°C (662-752°F), causing it to vaporize. The vapor enters a fractionating column, where it rises through trays or packing material.
As the vapor cools, different hydrocarbons condense at various levels of the column, depending on their boiling points. Lighter fractions condense at the top, and heavier ones condense at the bottom.
Some common fractions include:
- Petroleum gases (propane, butane) – top of the column, around 20-100°C.
- Naphtha (used for gasoline production) – 70-200°C.
- Kerosene (jet fuel) – 150-300°C.
- Diesel – 250-350°C.
- Heavy gas oils (lubricating oils, fuel oils) – 300-400°C.
- Residuum (asphalt, bitumen) – bottom of the column, remains as the heaviest fraction.
3. Conversion Processes
Some fractions, like heavy oils, may not be useful as-is. Refineries use conversion processes to break these heavy fractions into lighter, more valuable products. These processes include:
- Cracking: Large hydrocarbons are broken into smaller molecules. Types include thermal cracking (using heat) and catalytic cracking (using catalysts).
- Hydrocracking: Uses hydrogen and catalysts to break down heavier fractions into lighter, cleaner products like gasoline and diesel.
4. Treating and Reforming
- Treating removes impurities like sulfur, nitrogen, and metals from the fractions. This process uses techniques like hydrotreating or sweetening to make the products more environmentally friendly.
- Reforming converts the low-octane naphtha into high-octane gasoline by rearranging hydrocarbon molecules. It produces valuable byproducts like hydrogen.
5. Blending
The final products are blended to meet market specifications for fuels such as gasoline, diesel, and jet fuel. Additives may be mixed in to improve performance or reduce environmental impact.
6. Additional Processing Units
Coking: Used to process the heaviest residual oils, yielding lighter products and petroleum coke (used in industrial applications).
Isomerization and Alkylation: Produce higher-octane gasoline components by altering molecular structures.
Final Products
- Fuels such as Gasoline, diesel, kerosene, jet fuel, and fuel oil.
- Lubricants: Motor oils, greases.
- Petrochemicals are used to make plastics, synthetic rubber, and chemicals.
- Other byproducts: Asphalt, paraffin wax, sulfur.
This entire refining process transforms crude oil into usable products by separating it into various hydrocarbon chains with specific applications.
FAQ About Boiling Point and Separation of Petroleum Oil
What is crude oil?
Crude oil (petroleum) is a natural mixture of hydrocarbons formed from ancient plants and animals. It must be separated into useful fuels and products before use.
What is the boiling point?
The boiling point is the temperature at which a liquid changes into a gas. Different hydrocarbons in crude oil have different boiling points.
Why is the boiling point important in separating petroleum?
Fractional distillation separates crude oil because each fraction has a different boiling range. Components with lower boiling points vaporize and condense higher in the column; higher boiling point components condense lower down.
What is fractional distillation?
Fractional distillation is a process that separates crude oil into fractions (groups of hydrocarbons) based on differences in boiling points using a tall fractionating column with a temperature gradient.
Steps of Fractional Distillation of Crude Oil
What are the main steps in fractional distillation?
- Step 1: Heating the crude oil. Crude oil is heated in a furnace to about 350–400°C, turning most of it into vapor.
- Step 2: Entering the fractionating column. The hot vapors rise up the column, which is hot at the bottom and cooler at the top.
- Step 3: Condensation at different levels. As vapors rise, they cool and condense at different trays according to their boiling points.
- Step 4: Collection of fractions. Each fraction is collected separately (gases at the top, heavier oils near the bottom).
- Step 5: Further processing (optional). Some fractions are refined further by cracking, reforming, or treating to improve quality.
Main Fractions & Their Uses
What are the main fractions obtained from crude oil?
Common fractions include:
- Refinery gases (LPG) – cooking gas, heating.
- Petrol (Gasoline) – car fuel.
- Naphtha – petrochemicals, plastics.
- Kerosene – jet fuel.
- Diesel (Gas oil) – trucks, buses.
- Lubricating oil – engine oil.
- Fuel oil – ships, power stations.
- Bitumen (Residue) – roads, roofing.
Which fractions have low and high boiling points?
- Low boiling point fractions: gases, petrol (lighter molecules, more volatile).
- High boiling point fractions: fuel oil, bitumen (heavier molecules, less volatile).
Why do lighter fractions rise higher in the column?
Lighter hydrocarbons have lower boiling points, so they stay as vapor longer and rise higher before condensing in cooler regions.
Why is a temperature gradient important?
The temperature gradient allows each fraction to condense at a specific height, improving separation efficiency.
Is fractional distillation a physical or chemical process?
It is a physical separation process because no new substances are formed; components are separated based on boiling points.
Is fractional distillation harmful to the environment?
Refining can cause pollution if not controlled. Modern refineries use filters, scrubbers, and safety systems to reduce emissions and waste.
Why is petroleum separation important in daily life?
Because it provides essential fuels and materials for:
- Transportation.
- Electricity generation.
- Plastics and chemicals.
- Roads and construction.
Why can’t crude oil be used directly?
Crude oil is a mixture; it must be separated into useful fractions with specific properties.
What determines where a fraction condenses in the column?
Its boiling range and the temperature at that level of the column.
What happens to the residue at the bottom of the column?
The residue (bitumen) is collected at the bottom and used for roads, roofing, or further processing.
You can subscribe to Science Online on YouTube from this link: Science Online
Offshore Drilling advantages and disadvantages
Properties of fluids, Factors affecting density and pressure
Applications on the pressure at a point (Connected vessels, U-shaped tube & Mercuric barometer)