Properties and uses of some elements in the modern periodic table, How are elements used in daily life?
The modern periodic table organizes elements based on their atomic number and electron arrangement, which determine their physical and chemical properties. The uses of the elements in our lives depend on their properties. The elements are very important in our life, and we use them in many applications.
Elements
Metals such as Iron, Copper, and Aluminum are known for their strength, conductivity, and malleability, making them essential in construction, electrical wiring, and manufacturing. Nonmetals like Oxygen and Nitrogen have different properties, such as low conductivity and high reactivity, and they play vital roles in respiration, agriculture, and various chemical processes.
In addition, metalloids such as Silicon have properties between metals and nonmetals, making them crucial in electronics and computer technology. Some groups of elements have unique characteristics; for example, noble gases like Helium are very stable and used in balloons and cooling systems, while alkali metals such as Sodium are highly reactive and widely used in chemical industries. Overall, the periodic table helps us understand how elements behave and how they are used in everyday life, from technology to medicine.
Uses of some elements
Sodium is a metal, and it is a good conductor of heat. It is used in a liquid state to transfer the heat from inside the nuclear reactor to outside. This heat is used to obtain the vapour energy required to generate electricity.
Nitrogen gas is a non-metal element. The liquified nitrogen is used in the preservation of the cornea of the eye, due to the decrease in its boiling point.
Radioactive cobalt 60 is used in food preservation, because it radiates gamma rays which prevent the reproduction of the microbial cells, but they do not harm the human.
Silicon is a metalloid. Silicon slides are used in the manufacture of electronic devices such as computers, Because Silicon is a semiconductor, and its ability to conduct electricity depends on the temperature.
We can get rid of the gases of the undesirable odours inside the refrigerator by using a piece of coal that collects the gases on its surface.
FAQ about Elements in the Modern Periodic Table
1. What determines the properties of elements in the periodic table?
The properties of elements depend mainly on their atomic number and electron configuration, which influence how atoms bond and react with others.
2. What are the general properties of metals?
Metals are typically:
- Good conductors of heat and electricity.
- Malleable and ductile.
- Shiny (lustrous).
- Usually solid at room temperature (except mercury).
3. What are the common uses of metals?
- Iron: construction and manufacturing of steel.
- Copper: electrical wiring.
- Aluminum: aircraft, packaging, and utensils.
4. What are the general properties of nonmetals?
Nonmetals are usually:
- Poor conductors of heat and electricity.
- Brittle in solid form.
- Not shiny.
- Can exist as solids, liquids, or gases.
5. What are the uses of some nonmetals?
- Oxygen: respiration and combustion.
- Nitrogen: fertilizers and food preservation.
- Chlorine: water purification and disinfectants.
6. What are metalloids, and why are they important?
Metalloids have properties between metals and nonmetals. They are important in electronics. Example: Silicon is widely used in computer chips and solar panels.
7. How do noble gases differ from other elements?
Noble gases are very stable and unreactive because their outer electron shells are full. Example: Helium is used in balloons and cooling systems.
8. Why are alkali metals highly reactive?
Alkali metals have one electron in their outer shell, making them eager to lose it and form compounds. Example: Sodium reacts vigorously with water.
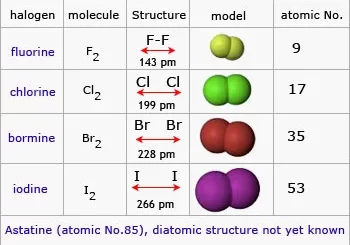
9. What are halogens and their uses?
Halogens are reactive nonmetals that form salts with metals. Example: Fluorine is used in toothpaste, and chlorine is used for disinfection.
10. How are transition metals useful?
Transition metals are strong and have multiple oxidation states. Example: Gold is used in jewelry and electronics, while Silver is used in photography and electrical contacts.
11. Why is carbon considered a special element?
Carbon can form a wide variety of compounds, making it essential for organic life and chemistry.
12. How are elements used in daily life?
Elements are used in:
- Medicine (oxygen, iodine).
- Industry (iron, aluminum).
- Technology (silicon, copper).
- Agriculture (nitrogen, phosphorus).
The modern periodic table organizes elements based on their properties, helping us understand how they behave and how they are used in everyday life—from construction and electronics to medicine and environmental protection.
You can follow science online on YouTube from this link: Science online
Description of the modern periodic table
General properties of halogens in the modern periodic table
General properties of the alkali metals in the modern periodic table