Metalloids, Difference between positive ion & negative ion, Why is Silicon the most famous metalloid?
Metalloids are semi-metallic elements that have the properties of both metals and nonmetals. It is difficult to know the metalloids from their outermost electrons due to the difference in the number of electrons in their valencies shells.
Metalloids
Metalloids occupy a unique diagonal territory on the periodic table, acting as a bridge between the distinct worlds of metals and nonmetals. These elements—which include silicon, boron, and arsenic—often have a physical appearance that mimics metals, typically displaying a lustrous or shiny surface.
However, they are chemically fragile and brittle, lacking the malleability that allows true metals to be hammered into sheets or drawn into wires. Because they sit on the “staircase” boundary, their behavior is highly context-dependent; they can act as insulators at low temperatures or under certain conditions, yet they become efficient conductors when heated or combined with specific impurities.
The most defining characteristic of metalloids is their role as semiconductors, a property that has made them the backbone of modern technology. Unlike metals, which conduct electricity freely, or nonmetals, which block it, metalloids allow for the precise control of electrical flow.
This unique ability is why silicon and germanium are indispensable in the manufacturing of microchips, transistors, and solar cells. Beyond electronics, their “in-between” nature allows them to form diverse chemical bonds, leading to their use in everything from flame retardants and glass strengthening to specialized medical treatments and pesticides.
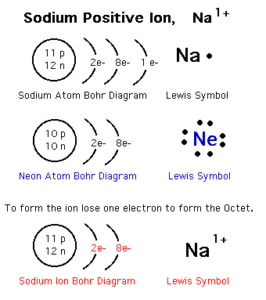
Positive ion
The positive ion (the cation) is the atom of a metallic element that loses one electron or more during the chemical reactions. The positive ion carries positive charges equal to the number of lost electrons.
Number of protons of the positive ion is more than that of its electrons. The electronic structure of the positive ion is similar to that of the nearest preceding inert gas. Number of energy levels in the positive ion is less than the number of energy levels in its atom.
Negative ion
Negative ion (the anion) is an atom of a nonmetallic element that gains one electron or more during chemical reactions. The number of electrons of the negative ion is more than that of its protons.
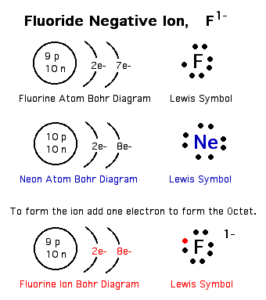
The number of energy levels in the negative ion is equal to the number of energy levels in its atom.
Negative ion carries negative charges equal to the number of gained electrons. You notice that the number of energy levels in the negative ion is equal to the number of energy levels in its atom. The electronic structure of the negative ion is similar to that of the nearest inert gas that follows.
FAQ about Metalloids
Metalloids are the “chameleons” of the periodic table, occupying the jagged line between metals and nonmetals. They are unique because they exhibit a blend of properties from both categories.
1. Which elements are considered metalloids?
While definitions vary slightly among scientists, the six commonly recognized metalloids are:
- Boron (B).
- Silicon (Si).
- Germanium (Ge).
- Arsenic (As).
- Antimony (Sb).
- Tellurium (Te).
2. What are their physical and chemical properties?
Metalloids are known for their “half-and-half” nature:
- Appearance: Most have a metallic luster (they look like metals), but they are brittle rather than malleable.
- Conductivity: They are semiconductors. They don’t carry electricity as well as metals, but they do it better than nonmetals. This makes them essential for modern electronics.
- Reactivity: Their chemical behavior depends on the element they are reacting with.
3. Why is Silicon the most famous metalloid?
Silicon is a semiconductor, meaning its ability to conduct electricity can be controlled. This property is the foundation of the entire computing industry—it’s the “Silicon” in Silicon Valley.
Positive Ions (Cations) vs. Negative Ions (Anions)
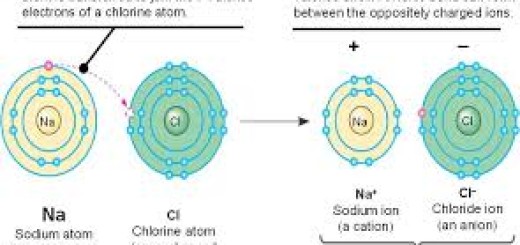
An ion is simply an atom that has gained or lost electrons, resulting in a net electrical charge. The difference between a positive and a negative ion comes down to the balance of protons (positive) and electrons (negative).
Positive Ions (Cations)
Cations are formed when a neutral atom loses one or more electrons from its outer shell. Because electrons carry a negative charge, losing them leaves the atom with more protons than electrons, resulting in a net positive charge.
This process typically happens to metals, which have few outer electrons and find it “easier” to give them away to achieve stability. When an atom becomes a cation, it physically shrinks in size because it has lost an electron cloud layer, and the remaining electrons are pulled tighter toward the nucleus.
Negative Ions (Anions)
Anions are formed when a neutral atom gains one or more electrons. By adding extra negative electrons to its structure, the atom now has more electrons than protons, resulting in a net negative charge.
This process is characteristic of nonmetals, which have nearly full outer shells and “prefer” to pull electrons from other atoms to fill the gaps. When an atom becomes an anion, it usually grows in size because the extra electrons repel each other, causing the electron cloud to expand.
Why do they form?
Atoms generally “want” a full outer shell of electrons to become stable (often referred to as the Octet Rule). Metals usually have 1–3 electrons in their outer shell, so it is easier to “throw them away” (becoming positive). Nonmetals usually have 5–7 electrons, so it is easier to “grab” a few more to fill the gap (becoming negative).
You can follow science online on YouTube from this link: Science online
Metallic and nonmetallic property, Acidic and basic property in the periodic table
Radius property, Ionization potential, Electron affinity and Electronegativity