The description of the modern periodic table, Why is the modern periodic table important?
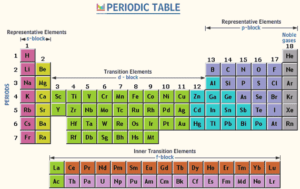
The modern periodic table consists of 7 horizontal periods and 16 vertical groups (18 vertical columns). The elements of the modern periodic table are classified into 4 blocks (s, p, d, and f).
The periods in the modern periodic table
The periods include elements of different properties, and they have the same number of energy levels.
By increasing the atomic number for the elements of the period, the atomic size decreases, the electronegativity increases, and the metallic property decreases till we reach the metalloid, then the nonmetallic property increases.
The groups in the modern periodic table
The groups include elements of similar properties, and they have the same number of electrons in the outermost energy level.
By increasing the atomic number of the elements in the groups, the atomic size increases, the electronegativity decreases, the metallic property increases in the groups that start with the metal, and the nonmetallic property decreases in the groups that start with the nonmetal.
The blocks in the modern periodic table
There are 4 blocks (s, p, d, and f). s-block elements are located on the left side of the periodic table, and they are arranged in two groups (1 A) and (2 A).
p-block elements are located on the right side of the periodic table, and they are arranged in six groups (3 A), (4 A), (5 A), (6 A), (7 A), and the zero group.
d-block elements are located in the middle of the periodic table, and they are arranged in eight groups (3 B), (4 B), (5 B), (6 B), (7 B), (8), (1 B), and (2 B).
d-block elements are known as transition elements. They appear to start from the period (4). All the groups in the d-block take the letter “B” except group (8), which consists of three vertical columns.
f-block elements are located below the periodic table. They include lanthanides and actinides.
The elements of the group (A) are located on the left and right sides of the table. The elements of group (B) are located in the middle of the table, and the new number of group (5 A) is (15), while that of the zero group is (18).
FAQ about the Description of the Modern Periodic Table
1. What is the modern periodic table?
The modern periodic table is a systematic arrangement of chemical elements based on their atomic number (number of protons), electron configuration, and recurring chemical properties. It organizes elements into rows and columns to show patterns in their behavior.
2. Who developed the modern periodic table?
The foundation was laid by Dmitri Mendeleev, but the modern version is based on the work of Henry Moseley, who arranged elements according to atomic number instead of atomic mass.
3. How are elements arranged in the modern periodic table?
Elements are arranged in:
- Periods (horizontal rows): There are 7 periods.
- Groups (vertical columns): There are 18 groups.
- Elements in the same group have similar chemical properties.
4. What is the modern periodic law?
The modern periodic law states that: “The physical and chemical properties of elements are periodic functions of their atomic numbers.”
5. What are periods in the periodic table?
Periods are horizontal rows. As you move from left to right, the atomic number increases, and the properties change gradually from metals to nonmetals.
6. What are groups in the periodic table?
Groups are vertical columns. Elements in the same group have the same number of valence electrons and show similar chemical behavior.
7. What are the main categories of elements?
Elements are classified into:
- Metals (left side).
- Nonmetals (right side).
- Metalloids (between metals and nonmetals).
8. What are blocks in the periodic table?
The periodic table is divided into blocks based on electron configuration:
- s-block.
- p-block.
- d-block (transition elements).
- f-block (lanthanides and actinides).
9. Why is the modern periodic table important?
It helps scientists:
- Predict properties of elements.
- Understand chemical reactions.
- Classify elements systematically.
10. What is the difference between Mendeleev’s and the modern periodic table?
- Mendeleev arranged elements by atomic mass.
- The modern table uses atomic number.
- The modern table explains periodic trends more accurately.
11. How does atomic number affect element properties?
Atomic number determines:
- Number of protons and electrons.
- Position of the element in the table.
- Its chemical behavior and reactivity.
12. What are periodic trends?
Periodic trends are patterns seen in the table, such as:
- Atomic radius.
- Electronegativity.
- Ionization energy.
These trends change regularly across periods and groups.
You can subscribe to Science Online on YouTube from this link: Science Online
Elements of p-block, General properties of group 5A elements (group 15)
The most famous elements in 5A group (Nitrogen properties, Preparation and compounds)
Transition elements & Economic importance of elements of the first transition series
The lanthanides and actinides in the modern periodic table
Modern periodic table and classification of Elements
Graduation of the properties of the elements in the modern periodic table