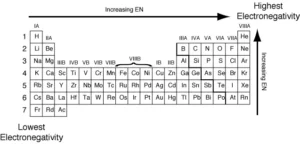
Graduation of Electronegativity of elements in modern periodic table, Why is electronegativity important?
Electronegativity is the ability of the atom in the covalent molecule to attract the electrons of the chemical bond towards itself. There is an inverse relationship between the atomic size of the atom of an element, and its electronegativity in the modern periodic table.
In periods in the modern periodic table. The electronegativity increases by increasing the atomic number (from left to right). The electronegativity for the elements of the same period is directly proportional to the atomic number.
In groups in the modern periodic table, the electronegativity decreases by increasing the atomic number (from up to down). The electronegativity for the elements of the same group is inversely proportional to the atomic number.
The inert gases do not have electronegativity as they do not enter into chemical reactions under normal conditions.
The difference in the electronegativity between the elements forming the compound molecules which determines the kind of bond between the elements, whether it is an ionic bond or a covalent bond.
The difference in the electronegativity between the elements forming the covalent molecules, which measures the polarity of these molecules, so the covalent compounds are classified into polar compounds and non-polar compounds.
The polar compounds are the covalent compounds in which the difference in the electronegativity between their elements is relatively high.
When the difference in the electronegativity between the elements of a covalent compound molecule equals zero, the bond in this molecule is called the pure covalent bond. As in the oxygen molecule and the hydrogen molecule.
FAQ About Electronegativity
1. What is electronegativity?
Electronegativity is the ability of an atom to attract shared electrons toward itself in a chemical bond.
2. What is meant by “graduation of electronegativity”?
It refers to the gradual change (trend) in electronegativity values of elements across periods and down groups in the modern periodic table.
3. How does electronegativity change across a period (left → right)?
Electronegativity increases from left to right. Reason: Increase in nuclear charge, Decrease in atomic size, and Stronger attraction for bonding electrons. Example: Sodium (Na) < Magnesium (Mg) < Chlorine (Cl)
4. How does electronegativity change down a group (top ↓ bottom)?
Electronegativity decreases down the group. Reason: Increase in atomic size, Increased electron shielding, Weaker attraction for bonding electrons. Example: Fluorine (F) > Chlorine (Cl) > Bromine (Br).
5. Which element has the highest electronegativity?
Fluorine (F) is the most electronegative element.
6. Which elements have the lowest electronegativity?
Alkali metals like Cesium (Cs) and Francium (Fr), They have a large atomic size and a weak attraction for electrons.
7. Why do noble gases usually not have electronegativity values?
Because they have complete outer electron shells. They rarely form bonds, so electronegativity is not commonly measured for them.
8. What factors affect electronegativity?
- Atomic size (smaller atoms → higher electronegativity).
- Nuclear charge (more protons → stronger attraction).
- Shielding effect (more inner electrons → lower electronegativity).
9. Why is electronegativity important?
Because electronegativity determines: Type of chemical bond (ionic or covalent), Polarity of molecules, and Chemical reactivity.
10. How can you remember the trend easily?
Simple rule:
- Across a period → increases
- Down a group → decreases
Think: “Top right = highest electronegativity”.
You can subscribe to Science Online on YouTube from this link: Science Online
Graduation of the properties of the elements in the modern periodic table
Attempts of elements’ classification
The description of the modern periodic table
Modern periodic table and classification of Elements
Chemical combination, Types of bonds (Chemical bonds & Physical bonds)