Graduation of metallic and nonmetallic property in the modern periodic table
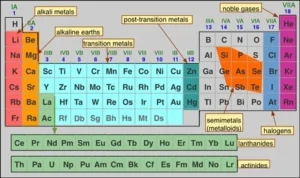
The modern periodic table is a systematic arrangement of elements based on their atomic number, which reveals clear patterns in their physical and chemical properties. One of the most important trends observed in this arrangement is the gradation of metallic and nonmetallic properties. These properties do not remain constant; instead, they change gradually as we move across periods and down groups, helping scientists understand the behavior of different elements.
Modern periodic table
Metallic and nonmetallic properties are closely related to an element’s ability to lose or gain electrons. As we move across a period from left to right, metallic character decreases while nonmetallic character increases. In contrast, moving down a group shows the opposite trend. Understanding these variations is essential for predicting reactivity, bonding, and the overall nature of elements in the periodic table.
In the modern periodic table, each period starts with a strong metal (in group 1 (1 A), where the metallic property decreases by increasing the atomic number, till we reach the metalloids.
Then the nonmetallic property appears, and increases by increasing the atomic number till we reach the strongest nonmetal (in group 17 (7 A) and ends in the inert gas (group zero).
By increasing the atomic number (within a period), the metallic property decreases, while the nonmetallic property increases.
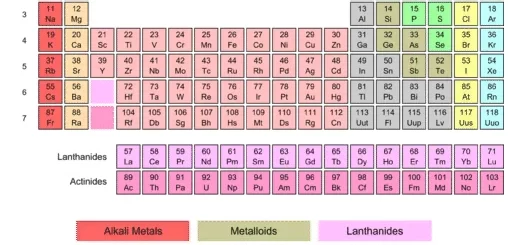
In metallic groups, the metallic property increases gradually as we go from top to bottom, as in group (1 A) (because the atomic size increases).
When you look within group (1 A) in the modern periodic table, you will notice the graduation of the metallic property. Cesium (Cs) is the most metallic element in the group (1 A), and lithium (Li) is the least metallic element in the group (1 A).
In nonmetallic groups, the nonmetallic property decreases gradually as we go from top to bottom, as in group (7 A) (due to the decrease in electronegativity).
When you look within group 7 A (17) in the modern periodic table, you will notice the graduation of nonmetallic property, Flourine (F) is the strongest nonmetal element in group (7 A). and Iodine (I) is the least nonmetal element in the group (7 A).
When you look at the modern periodic table, you notice that there is a direct relationship between the atomic size of an element and its metallic property. There is a direct relationship between the electronegativity of an element and its nonmetallic property.
FAQ about Gradation of Metallic and Nonmetallic Properties
1. What is meant by gradation in the periodic table?
Gradation refers to the gradual change in properties of elements as we move across a period or down a group in the modern periodic table.
2. What are metallic properties?
Metallic properties include:
- Ability to lose electrons.
- Good conductivity of heat and electricity.
- Malleability and ductility.
- Shiny (lustrous) appearance.
3. What are nonmetallic properties?
Nonmetallic properties include:
- Ability to gain electrons.
- Poor conductivity.
- Brittle nature.
- Dull appearance.
4. How do metallic properties change across a period (left to right)?
Metallic properties decrease from left to right across a period because atoms hold their electrons more strongly.
5. How do metallic properties change down a group?
Metallic properties increase down a group because atoms become larger and lose electrons more easily.
6. How do nonmetallic properties change across a period?
Nonmetallic properties increase from left to right across a period due to the increased attraction for electrons.
7. How do nonmetallic properties change down a group?
Nonmetallic properties decrease down a group because atoms become larger and less able to attract electrons.
8. Why do these trends occur?
These trends occur due to:
- Increase in atomic number.
- Changes in atomic size.
- Variation in nuclear charge and electron attraction.
9. Where are metals and nonmetals located in the periodic table?
- Metals are found on the left and center of the table.
- Nonmetals are found on the right side.
- Metalloids lie in between.
10. What is the significance of these trends?
Understanding these trends helps in:
- Predicting chemical behavior.
- Determining the reactivity of elements.
- Explaining bonding and reactions.
You can subscribe to Science Online on YouTube from this link: Science Online
Metallic & nonmetallic property, Acidic & basic property in the periodic table
Atoms components, Rutherford and Bohr’s Atomic Models
Modern periodic table and classification of Elements
Radius property, Ionization potential, Electron affinity & Electronegativity
Graduation of the properties of the elements in the modern periodic table