Carbohydrates importance, Types of Isomerism, Monosaccharides and Disaccharides
Carbohydrates provide your body with energy. Most carbohydrates are digested and broken down into glucose before entering the bloodstream. Cells can convert carbohydrates into the fuel molecule ATP through a process called cellular respiration. Most cells in the body can produce ATP from several sources, including dietary carbohydrates and fats.
Carbohydrates
They are organic substances composed of carbon, hydrogen, and oxygen. There are three main types of carbohydrates: starches, fiber, and sugars. Starches are referred to as complex carbohydrates. They are found in grains legumes and starchy vegetables like potatoes and corn. Sugars are known as simple carbohydrates. There are natural sugars in vegetables, fruits, milk, and honey. Added sugars are found in processed foods, syrups, sugary drinks, and sweets.
Classification of carbohydrates
- Monosaccharides (simple sugars): These are the simple units i.e can not be hydrolyzed into a simpler form.
- Oligosaccharides (oligo = few): These are carbohydrates containing from two to ten monosaccharide units. e.g. disaccharides (2 units), trisaccharides (3 units)…..etc.
- Polysaccharides (poly=many): Also known as glycans, they are composed of more than ten monosaccharide units e.g. starch, glycogen, cellulose…..etc.
Monosaccharides
Classification of monosaccharides
1- Monosaccharides are classified according to the number of carbon atoms: Trioses, tetroses, pentoses, hexoses & heptoses.
2- According to the characteristic carbonyl group (aldehyde or ketone group):
Aldo sugar (aldoses): monosaccharides containing aldehyde group, e.g.
- Glyceraldehyde
- Ribose: is a structural element of ribonucleic acid (RNA), coenzymes (NAD, NADP, …), and high energy compounds (ATP, GTP, …).
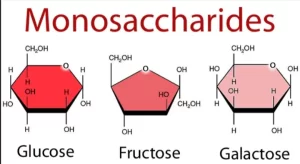
- Glucose: blood sugar; it is utilized by all cells, and called dextrose because it is dextrorotatory.
- Galactose: is a component of mucopolysaccharides and galactolipids.
Keto sugars (ketoses): monosaccharides containing ketone group e.g.
- Fructose: is present in semen as a nutrient for sperms.
Ring structure of monosaccharides
- Monosaccharides of five or more carbon atoms are present in ring forms.
- The ring formation includes condensation of the carbonyl group (C-1 in aldoses, and C-2 in ketoses) of the sugar with one of the alcohol groups of the same sugar forming a closed ring structure.
- The carbonyl carbon atom in the cyclic form will be called the anomeric carbon and it can exist in two different forms called α (with the hydroxyl group to the right) or β (with the hydroxyl group to the left).
- In Haworth configuration, all groups to the right of carbons are oriented down while all groups to the left of carbons are oriented up except those around carbon 5, the reverse orientation occurs.
Asymmetric carbon atom
An asymmetric carbon atom is a carbon atom attached to four different atoms or groups. Any compound having asymmetric carbon(s) will possess isomers. Isomers are compounds that have the same molecular weight, same percentage composition, and differ in their physical and chemical properties.
Important types of isomerism
1- D and L isomers
They are two isomers; one of them is the mirror image of the other e.g. D and L glyceraldehydes & D and L glucose. Most of the monosaccharides occurring in mammals are of the D configuration.
2- Epimers
Two monosaccharides differ only in the configuration around one specific carbon atom (not the anomeric), the D-glucose & D-mannose are the epimers with respect to carbon atom 2, and D-glucose & D-galactose are the epimers with respect to carbon atom 4.
3- Aldose-Ketose isomerism
Two monosaccharides have the same molecular formulae but differ in their functional groups, since one has an aldehyde group and the other has a ketone group, glucose and fructose are examples of this type of isomerism.
4- α and β anomers
α and β – anomers are the isomers of monosaccharides that differ from each other only in the configuration about the anomeric carbon.
Derivatives of monosaccharides
1- Sugar acids: these are oxidation products of monosaccharides.
Biologically important sugar acids:
- Uronic acids are obtained by oxidation of the last carbon e.g. D-glucose yields D- glucuronic acid
(it is important as a component of mucopolysaccharide and also in detoxication processes). - The L-ascorbic acid (vitamin C) is another important naturally occurring sugar acid.
2- Sugar alcohols: These are reduction products of monosaccharides. e.g. ribose gives ribitol, glucose gives sorbitol, mannose gives mannitol, and galactose gives galactitol, while fructose gives rise to either sorbitol or mannitol. Besides those sugar alcohols (itols), myoinositol is a cyclic sugar alcohol that occurs in nature and is the important information of phosphatidylinositol.
3- Deoxysugars: In deoxysugars, a hydroxyl group has been replaced by a hydrogen atom, the most abundant naturally occurring deoxyribose are: 2-deoxyribose, a constituent of DNA.
4- Aminosugars: They are monosaccharides in which the hydroxyl group at carbon 2 is replaced by an amino group (NH2) which may be acetylated, e.g. D-glucosamine, D-galactosamine.
5- Amino sugar acids (e.g. Neuraminic acids) are nine-carbon amino sugar derivatives consisting of 6-carbon aminosugar linked to 3-carbon acid; the amino group is usually acetylated. E.g. Neuraminic acid is formed of mannosamine linked to pyruvic acid, sialic acid is N-acetyl neuraminic acid which is present in glycolipids (Gangliosides).
6- Glycosides: Glycosides are compounds formed from condensation between:
- A sugar and another sugar e.g. disaccharides or higher oligo and polysaccharides.
- Sugar and an aglycone (non-sugar) e.g. Nucleosides = pentose + nitrogenous base, these are components of nucleotides and nucleic acids.
Disaccharides
Disaccharides have two monosaccharides, the same or different, linked by glycosidic linkage e.g.:
1- Maltose (Malt sugar)
Chemistry: Maltose contains two D-glucose residues, in pyranose form, linked by an α-1-4 glycosidic bond.
Sources of maltose:
- Germinating cereals and malt.
- The intermediate product of the action of amylases on starch.
2- Lactose (Milk sugar)
Chemistry: D-glucose and β-D galactose, in pyranose form, linked by β -1,4-galactosidic linkage.
Sources: It is only found in milk.
3- Sucrose (Cane sugar = Table sugar = Beet sugar)
Chemistry: It is a disaccharide of β-D fructofuranose and α-D-glucopyranose linked by β-2,1 fructosidic linkage or α-1,2 glucosidic linkage. The anomeric carbon atoms of the two hexoses are linked to each other, thus, sucrose contains no free anomeric carbon atom.
You can subscribe to Science Online on YouTube from this link: Science Online
Carbohydrates, Polysaccharides, Proteoglycans, Glycoproteins & Glycosaminoglycans
Connective tissue cells types, function & structure, Resident cells & Transient cells
Integumentary system, Skin importance, layers, types & function