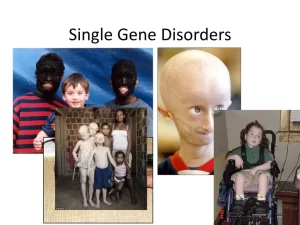
Polygenic inheritance vs Single gene inheritance, Non-mendelian inheritance and Genomic imprinting
Mutations in the DNA sequence of a specific gene cause a single-gene disorder, The DNA changes affect the product that the gene codes for—usually a protein—causing it to be altered or missing, Some genetic disorders are so severe that children with them are extremely sick or cannot survive after birth, Others are easy to manage, and with proper care, people with them have very fulfilling lives.
Single gene inheritance
A number of single-gene disorders do not follow classical patterns of inheritance, Recently several molecular mechanisms have been recognized that can account for this.
Genomic imprinting
Normally some genes show differences in expression according to the parent of origin, where inactivation of these genes occurs most likely by methylation during gamete formation and persists permanently in a such inactive form and passes on to offspring in relation to parent sex imprint. In such a case, the allele derived from the other parent will be the only active allele copy of such gene.
Hence different clinical features can result depending on whether the defective gene is inherited from the mother or the father. This has been observed in two clinical syndromes associated with an imprinted region on chromosome 15 (15q11-q13). Mental handicap with obesity (eg Prader Willi Syndrome) occurs if the mutant gene was inherited from the father (it results from the interstitial deletion of the long arm of chromosome 15 derived from the father.
Other causes may be maternal uniparental disomy. Whereas, other severe disabling disorder of severe mental disability and convulsions (Angelman Syndrome) occurs if the defective gene is inherited from the mother (occurs with interstitial deletion of the long arm of chromosome 15 is derived from the mother. Or it may result from paternal uniparental disomy and be shown by DNA analysis).
Uniparental disomy is a condition in which both pairs of the homologous chromosomes are derived from the same parent, by a mechanism of trisomy rescue.
Mitochondrial inheritance
The disorder is transmitted to both sex, but is only transmitted through females due to exclusive maternal contribution of mitochondrial DNA to offspring. The proportion of affected offspring is variable and the clinical picture within a family is variable, this is due to the proportion of normal and mutant copies of mitochondrial DNA passed on in the ova from the mother to the offspring, Mitochondrial inherited disorders include a number of rare disorders usually affecting high energy consuming organs. They are usually characterised by an unusual combination of myopathic and/or neurologic features.
Examples include:
- Leber’s optic atrophy (Hereditary blindness resulting from mutation of the mitochondrial DNA gene involved in detoxification of cyanide).
- Deafness (antibiotic-induced deafness, some forms of progressive nerve deafness).
- Diabetes mellitus (some familial forms).
- A mitochondrial variant of cardiomyopathy.
Unstable (dynamic) or expanding triplet repeats Mutations
This mutation results from expanding the number of trinucleotides repeats within particular genes. Clinical examples include:
- Fragile X Syndrome: mental retardation is more common in males than females. Fragile S is inherited as an X-linked disorder which presents more in males. Fragile X Syndrome results from the expansion of CGG repeats within the Fragile X gene (normal number is 50), premutation is between 50-200 and the full mutation is >200. The severity of the phenotype correlates with the size of the repeat.
- Myotonic Dystrophy: the severity depends on the expansion size. The DNA expansion occurring in the mother usually results in severe phenotype.
- Huntington Disease: the larger the number of repeats, the earlier the expression of the disease (juvenile-onset HD). The large expansions are usually transmitted from the father.
These disorders show anticipation as a result of the dynamic nature of this type of mutation which increases in successive generations.
Multifactorial/ Polygenic Disorders
This type of inheritance is determined by the contribution of several genes of small effect but additive together with environmental influence. The sum of these genetic and environmental factors determines a person’s susceptibility to be affected by a particular disorder. This category of genetic disorders involves the most common medical disorders of adulthood such as diabetes, hypertension, cardiovascular diseases, Alzheimer’s disease, congenital malformations, and cancer.
Phenotype of multifactorial trait/disease
Depending on the type of phenotype:
Qualitative Trait: the clinical condition is either present or absent.
Quantitative Trait: the clinical condition with a measurable quantity that differs among individuals (measurable). For example: blood pressure, cholesterol level, height etc. Variation among individuals is due to differences in genotype as well as environmental factors.
This determines the heritability of a trait: the amount of variations observed in a certain population that is determined by individual genetics factors. Some populations are at more risk of certain disorders due to genetic background.
Recurrence Risk of polygenic disorders: The probability that a multifactorial genetic disorder in at least one family member will occur again in another family member is based on empiric risk and is calculated as 2-4%. There is an increased individual risk if:
- Multiple members of a family are affected.
- Proband is of rarely affected sex (e.g. male breast cancer).
- Close relationship to proband; recurrence risk is high in close relatives and decreases in distant relatives.
- Severe or early onset of the proband.
- Consanguinity.
- The recurrence risk is based on the observation of large data of the affected and unaffected in family studies and is specific to a certain population, “Empiric risk”.
You can subscribe to science online on YouTube from this link: Science Online
You can download Science Online application on Google Play from this link: Science Online Apps on Google Play
Cancer genetics, Genetic defects in tumours, Oncogene activation and Examples of hereditary cancer
Chromosomal disorders, structure, classification, and Types of numerical chromosomal abnormalities
Genes, Chromosomes, Proteins, Bacteriophages & Quantity of DNA in the cells
Nucleus components, function, diagram & classification of chromosomes
Cells types, Chromosomes, Cell division, Phases of mitosis division & Liver Transplantation
Packaging of DNA, Genome, chromosomal proteins, DNA in Prokaryotes and Eukaryotes