Control of insulin secretion, diabetes mellitus, Glucagon, Somatostatin and features of DNES
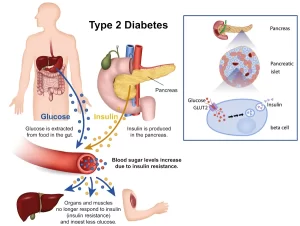
Insulin is a peptide hormone made in your pancreas, a gland located behind your stomach, It enables your body to use glucose for energy, It regulates the metabolism of carbohydrates, fats & protein by promoting the absorption of glucose from the blood into the liver, fat and skeletal muscle cells, Insulin allows your body to store glucose in your liver, fat, and muscles, the pancreas controls the amount of glucose in your bloodstream at any given moment.
Control of insulin secretion
- Simulation of insulin secretion by blood glucose: The primary stimulus for increased insulin secretion is an increase in blood glucose concentration. At the normal fasting level of blood glucose of 80-90 mg/dl, the rate of insulin secretion is minimal. If the blood glucose concentration is suddenly increased to a level two to three times normal, insulin secretion increases. Glucose penetrates the beta cells directly and its rate of entrance is unaffected by insulin when the level of glucose in the pancreas is increased, insulin secretion increases.
- Amino acids potentiate the glucose stimulus for insulin secretion, e.g. arginine & lysine.
- An increase of intracellular potassium decreases insulin secretion.
- Gastrointestinal hormones: Gastrin, secretin, cholecystokinin, and gastric inhibitory peptide may cause a moderate increase in insulin secretion.
- Other hormones: Include glucagon, growth hormone, cortisol, and to a lesser extent progesterone and estrogen, Prolonged secretion of any one of them in, large quantities can lead to the exhaustion of the beta cells of the islets of Langerhans and cause diabetes.
- Autonomic nervous system: The autonomic innervation of the pancreas is involved in the regulation of insulin secretion, Stimulation of the vagus nerve causes increased insulin secretion. However, stimulation of the sympathetic nerves to the pancreas inhibits insulin secretion. The inhibition is produced by released norepinephrine acting on α2 adrenergic receptors.
Effects of insulin deficiency (diabetes mellitus)
Deficiency of insulin causes disturbances in carbohydrate, fats, and protein metabolism.
A) Disturbance in carbohydrate metabolism:
- Polyuria is due to the osmotic diuretic effect of glucose in the kidney tubules. Loss of glucose in the urine of the diabetic person occurs when blood glucose level rises over 180 mg/dL, which is called the blood threshold for the appearance of glucose in the urine. The loss of glucose in the urine causes osmotic diuresis because the osmotic effect of glucose in the tubules prevents tubular reabsorption of fluid. Large amounts of water are lost and this can lead to dehydration.
- The resulting dehydration activates the mechanisms regulating water intake leading to polydipsia.
- The failure of glucose (and protein) utilization by the body causes loss of weight and a tendency toward polyphagia.
- There is urinary loss of sodium and potassium which leads to dehydration, hypovolemia, and hypotension.
B) Disturbances in protein metabolism:
- Protein synthesis stops so the rate of growth is decreased especially in diabetic children and it causes asthenia in adults.
- Catabolism of proteins increases.
- Most of the excess amino acids are either used directly for energy or for gluconeogenesis.
- Degradation of the amino acids increases urea excretion in the urine.
- Poor resistance to infection due to a decrease of protein and the sugar-rich body fluids are good culture media for microorganisms.
C) The disturbance in fat metabolism which leads to:
- The hydrolysis of the stored triglycerides, releases large quantities of fatty acids and glycerol into the circulating blood.
- Free fatty acids then become the main energy substrate used by essentially all tissues of the body besides the brain.
- The excess of free fatty acids in the blood causes rapid diffusion of fatty acids into the liver cells.
- The liver forms triglycerides, phospholipids, and cholesterol from excess fatty acids, This leads to the rapid development of atherosclerosis in persons with serious diabetes and fatty liver.
Glucagon
Glucagon is a single-chain polypeptide, consisting of 29 amino acids, Glucagon contains no cysteine residues and thus has no disulphide bond.
Metabolism of glucagon
The kidney and the liver play an important role in the metabolic clearance of glucagon. The liver rapidly inactivates the hormone by enzymatic cleavage of amino acids from the N terminal and of the hormone.
Mechanism of glucagon action
After binding to a specific hormone receptor on the liver plasma membrane, glucagon is internalized and degraded thereafter. The most accepted scheme by which glucagon activates cAMP production is through activation of adenylcyclase enzyme converting ATP to cAMP, which by activating cAMP-regulated protein kinase, mediates the physiologic effect of the hormone.
The effect of glucagon-like epinephrine and norepinephrine on cAMP is mediated through G protein. the G protein is formed of three subunits α, β, and γ. In the resting phase (i.e.in the absence of hormone) the G protein α subunit binds to GDP, and the hormone receptor, G protein, and adenylcyclase are not linked together. When the G protein interacts with the hormone-receptor complex, the affinity of the α subunit to GDP is decreased, but the affinity for GTP is increased.
Thus the G protein releases bound GDP and picks up a molecule of GTP. This allows the α subunit to bind transiently to adenylatecyclase switching on its activity. cAMP, in turn, converts phosphorylase enzyme to its active form and glycogen synthase to its inactive form.
Actions of glucagon
1- On CHO metabolism
Glucagon is a hyperglycemic hormone. It raises blood sugar levels by increasing:
- Breakdown of glycogen (glycogenolysis) in the liver but not in the muscle.
- Gluconeogenesis from available amino acids in the liver.
2- On lipid metabolism:
- Glucagon is a potent lipolytic agent. It increases adipose cell cAMP levels and this activates the hormone sensitive lipases
- It increases ketone body formation due to its lipolytic effect, The increased free fatty acids can be metabolized for energy or converted to ketone bodies.
3- It increases metabolic rate because it increases the hepatic deamination of amino acids.
4- It stimulates the secretion of GH insulin and pancreatic somatostatin.
Regulation of glucagon secretion
- A decrease in the blood glucose concentration increases glucagon secretion and increases the blood glucose concentration to hyperglycemic levels decreases glucagon secretion.
- High concentrations of amino acids after protein meal stimulate glucagon secretion. Arginine and phenylalanine are potent stimulators of glucagon secretion and both are used as tests for evaluation of alpha cell function.
- CCK and gastrin increase glucagon secretion, but secretin inhibits it.
- During exercise, the blood concentration of glucagon often increases to increase glucose production by the liver.
- Glucagon secretion is increased by sympathetic stimulation to the pancreas.
Somatostatin
It is secreted by the delta cells of the islets of Langerhans. Somatostatin is a polypeptide. It is the same chemical substance as growth hormone inhibitory hormone that is secreted in the hypothalamus, All factors related to the ingestion of food will stimulate somatostatin secretion, These include:
- Increased blood glucose.
- Increased amino acids.
- Increased fatty acids.
- Increased concentration of several of the gastrointestinal tract hormones in response to food intake.
Somatostatin has multiple inhibitory effects as follows:
- Somatostatin acts within the islets of Langerhans themselves to depress the secretion of both insulin and glucagon.
- Somatostatin decreases the motility of the stomach, the duodenum and the gallbladder.
- Somatostatin decreases both secretion & absorption in the gastrointestinal tract.
So, the principal role of somatostatin is to cause slowing of the assimilation of food from the gut.
Diffuse neuro-endocrine system (DNES)
It includes single or small groups of endocrine cells with diffuse distribution in different tissues and organs of the body. These cells secrete different hormones, but usually peptides; as they have the capacity to synthesize monoamines by the uptake of amine precursors and their decarboxylation, they are also “APUD cells” (Amine Precursor Uptake and Decarboxylation).
These hormones act in a paracrine (affect activities of neighboring cells). autocrine (on the cell that secreted the hormone itself) or endocrine mode and their function are regulated by nervous control.
Histological features of DNES
These cells are not well seen by routine H&E sections; however, they can be specifically visualized by LM after staining with chromium or silver salts. This provides them alternative names as chromaffin cells and argentaffin cells. More accurately, they are visualized by immunohistochemical staining using antibodies against their peptide hormone products.
By EM they are pyramidal in shape with a narrow apex and a wide base. They have the ultrastructural features of polypeptide secreting cells (rER, mitochondria, Golgi apparatus, and secretory granules) but not in abundance denoting their slower rate and a small amount of secretion. Golgi apparatus is infranuclear and secretory granules are basal to allow the secretion to pass to the underlying fenestrated capillaries. They are of two morphological types:
- Open type: in which the apex carries a tuft of long microvilli extending into the lumen of the organ.
- Closed type: where the cellular apex is covered by other epithelial cells.
Distribution of DNES
They are present mainly in the gastrointestinal tract (entero-endocrine cells) and respiratory system. Members of DNES of the endocrine system include:
- Supracptic and paraventricular hypothalamic nuclei produce oxytocin and vasopressin hormones.
- Parafollicular cells of the thyroid gland.
- Chief cells of the parathyroid gland.
- Chromaffin cells of the suprarenal medulla.
- Cells of islets of Langerhans in the pancreas.
You can subscribe to Science Online on YouTube from this link: Science Online
Histology of pancreas, Structure of islets of Langerhans, Insulin function and Metabolism
Cortisol effects, Cause of melanin deposition, Adrenal androgens and Adrenogenital Syndrome
Glucocorticoid (cortisol) function, Effects of cortisol on skeletal muscle and circulatory system
Calcium metabolism, Calcitonin function, Vitamin D3 and Biosynthesis of calcitriol
Endocrine system structure, function, disorders, Endocrine Glands and Hormones types