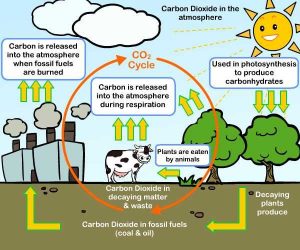
What are the sources of carbon dioxide gas?
Carbon dioxide gas is very necessary for the photosynthesis process of green plants to make their own food, and to build their bodies, It is a chemical compound which its molecule consists of one carbon atom (C) linked with two oxygen (O) atoms and it represents 0.03 % of the volume of the atmosphere.
Sources of carbon dioxide gas
The volcanic eruptions release magma, ash, dust & gases from deep below the Earth’s surface, one of the gases released is carbon dioxide, and the most common volcanic gases are water vapor, carbon dioxide, and sulfur dioxide.
Carbon dioxide gas increased due to human activities such as the burning of oil, coal and natural gas, burning of fossil fuels, clearing of forests, and some industrial processes such as cement manufacturing.
Carbon dioxide gas is produced from the combustion of fuel in the factories and means of transportation, and the largest human source of carbon dioxide emissions is from the combustion of fossil fuels.
Carbon dioxide gas is produced from the combustion of organic materials such as wood, coal, oil, gasoline, and tobacco (the material of cigarettes).
From the factors that increase the percentage of carbon dioxide gas in air is the respiration process of the human, the animals, and the plants, Carbon dioxide gas is produced from decreasing the green areas and removing the forests.
There are industrial processes that produce carbon dioxide gas as the production and consumption of mineral products such as cement, the production of metals such as iron and steel, as well as the production of chemicals and petrochemical products.
Cement production produces the most amount of carbon dioxide amongst all industrial processes, During creating the main ingredient in cement, calcium oxide, limestone is chemically transformed by heating it to very high temperatures, This process produces large quantities of carbon dioxide as a byproduct of the chemical reaction.
Carbon dioxide gas is produced from steel production, if you want to create steel, iron is melted and refined to lower its carbon content, and oxygen is combined with carbon in iron to form carbon dioxide gas.
Limewater is used to detect the presence of carbon dioxide gas, it turns into milky (turbid) when carbon dioxide passes through it due to the formation of a chemical substance called calcium carbonate which is insoluble in the water.
The fossil fuels are used to create the chemicals and petrochemical products which leads to carbon dioxide emissions, and the industrial production of ammonia and hydrogen most often uses natural gas or other fossil fuels as a starting base, creating carbon dioxide in the process.
The petrochemical products like the plastics, the solvents, and the lubricants are created using petroleum, and these products evaporate, dissolve, or wear out over time releasing even more carbon dioxide during the product’s life.
The properties of carbon dioxide gas
The importance and uses of carbon dioxide gas
What are the disadvantages of carbon dioxide?
Carbon monoxide emissions, sources, effects, uses, poisoning symptoms
Air pollution causes, types, effects, solutions & How to prevent air pollution
Dangers and bad effects of burning plastics and rubber on humans and global warming



It is very informative