Off-target effect of drugs types, Role of Precision Medicine and Pharmacogenetics
Years ago, patients were prescribed drugs for a certain disease blindly without prior genetic testing, but with the advancement of genetics medicine, and making use of the human genome project results, the term of precision medicine or personalized medicine and targeted therapy emerged.
Intended learning objectives
- Compare and contrast traditional and precision medicine.
- Differentiate terms of pharmacogenetics, pharmacogenomics and personalized medicine.
- Define xenobiotic genes and their role in drug metabolism.
- Identify the principal of tailoring therapy.
- Mention examples of personalized medicine based on genotyping in current practice.
If we compare traditional medicine and precision medicine, the earlier focuses on the use of drugs based on the average response of a group of patients while the latter involves the collection of extensive data on genetics, environment, and lifestyle to tailor the use of drugs to each patient’s genetic makeup.
Traditional medicine includes chemotherapy, surgery, & radiotherapy while precision medicine adopts genetic analysis to decide the expected response to a chemotherapeutic of choice. It uses the genomic data of the patient to maximize drug efficiency and minimize side effects.
Pharmacogenetics/pharmacogenomics, as the name implies, is defined as the study of genetic variations that cause a variable drug response among different individuals. It studies the genetic polymorphism of drug transporters, drug-metabolizing enzymes, and drug receptors.
The difference between pharmacogenetics and pharmacogenomics is that pharmacogenetics focuses on single drug-gene interaction while pharmacogenomics is broader and searches for genome-wide association studies (GWAS).
Pharmacokinetics includes (drug absorption, distribution, metabolism and elimination) and all of these steps are controlled by genes encoding proteins undertaking these functions. Pharmacodynamics is the effect of the drug exerted on organs and tissues of the body.
The liver is the site where drugs are handled. Some of the prescribed drugs are directly acting (need no metabolic conversion), the cytochrome oxidase enzymes (CYP) in mitochondria add or remove a hydroxyl or carboxyl group from the drug. This is called phase I metabolism. If the drugs need further chemical reactions, other enzymes undertake the metabolic conversion of the drug into an active compound, such as glutathione s methyl transferase enzyme (GSMT). This is called phase Il metabolism.
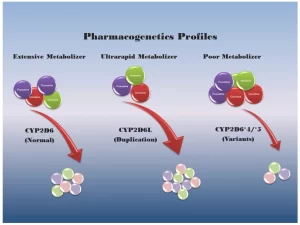
Slow metabolizers: express enzymes that detoxify drugs over a long period, therefore, the drug dose should be reduced to avoid drug side effects, while rapid metabolizers: express enzymes that detoxify drugs rapidly, hence the drug dose should be increased to give a desired therapeutic effect. The difference between both lies in the genes encoding the enzyme (SNPs)
CYP2D6 gene encodes an enzyme metabolizing a drug with selective serotonine release inhibitor (example prozac) to treat anexiety & depression, Rapid metabolizers need a larger dose of antipsychotic drug to exert good response while slow metabolizers need a smaller dose of the drug to avoid side effects
Off-target effect of drugs
Patients with uric acid, and kidney stones are treated with allopurinol (also used to treat hyperuricemia or gout), in a minority of patients with the HLA-B variant will have serious side effects causing Steven Johnson syndrome (10% fatality) so the genetic test for this variant is important to avoid serious consequences while using allopurinol.
Targeted therapy: is the use of a monoclonal antibody therapy targeting a certain molecule in a certain signal transduction pathway that is altered by a genetic mutation in certain cancers, for example, Trastazumab (herceptin) to treat her2neu gene amplification in breast carcinoma.
KIT gene is a proto-oncogene that encodes a signaling protein called a receptor tyrosine kinase. This protein transmits extracellular signals to activate pathways that promote the growth and survival of the cell.
Imatinib (Gleevec) is a drug, which targets some receptor tyrosine kinases, including c-KIT, and is often highly effective in treating gastrointestinal stromal tumours (GIST) with c-KIT driver mutations.
Warfarin is an anticoagulant used preventatively in individuals with atrial fibrillation, deep vein thrombosis or pulmonary embolism, It is particularly important to ensure that the levels of the drug remain in the therapeutic range, because too low a dose will be ineffective, and too high a dose can lead to bleeding.
The CYP2C9*2 and CYP2C9*3 alleles are present in European populations, Each variant allele produces CYP2C9 proteins with a single amino acid change. These changes to the DNA sequence can be described as a missense variant, The rates of warfarin metabolism by the protein encoded by the different CYP2C9 alleles vary, The warfarin metabolism rate of the proteins encoded by CYP2C9*2 and the CYP2C9*3 alleles are decreased by 30-40% and 80-90%, respectively, compared to the common version of the protein, encoded by CYP2C9*1.
This means that the warfarin dose should be adjusted according to each individual’s variant alleles, Patients with CYP2C9*3/CYP2C9*3, should take a low dose of warfarin to avoid side effects.
Warfarin acts by inhibiting the activity of the VKORC1 protein, which helps to produce functional clotting factors, There is a variant in the VKORC1 gene that necessitates lowering the dose of warfarin required for treatment, and individuals with this variant have an increased risk of bleeding when they are treated with warfarin, This variant is found in 1639 base pairs upstream of the translational start site, Therefore, this variant decreases the expression of the VKORC1 gene that’s why genetic testing is important before prescribing warfarin to these patients.
The environmental factors can also play a role in determining warfarin dosing, VKORC1 requires the cofactor called vitamin K to form active blood clotting factors, so increasing levels of vitamin K in the body leads to increased production of blood clotting factors, Leafy greens, such as kale and spinach, are rich in vitamin K, If someone on a consistent therapeutic dose of warfarin who typically does not eat a lot of leafy greens suddenly eats a giant kale salad, is at an increased risk of blood clotting.
You can subscribe to science online on Youtube from this link: Science Online
You can download Science Online application on Google Play from this link: Science Online Apps on Google Play
Methods of diagnosis of genetic variants, Ethical and social issues related to genetic testing
Genetics variants effects, Types of polymorphisms and disease-causing mutations
Polygenic inheritance vs Single gene inheritance, Non-mendelian inheritance and Genomic imprinting
Chromosomal disorders, structure, classification, and Types of numerical chromosomal abnormalities
Genes, Chromosomes, Proteins, Bacteriophages & Quantity of DNA in the cells
Packaging of DNA, Genome, chromosomal proteins, DNA in Prokaryotes & Eukaryotes