Protein Classification, Globular and Fibrous protein, Simple, Compound and Derived proteins
Protein is used to build bones, muscles, cartilage, and skin, Hair & nails are comprised mostly of protein, The body uses protein to build & repair tissues. Red blood cells contain a protein that carries oxygen throughout the body, This helps supply your body with the nutrients it needs. It is used in hormone regulation during the transformation & development of cells during puberty. It is used in making enzymes, which helps in food digesting and making new cells.
Classification of proteins
I. According to shape:
On the basis of axial ratios of proteins (the ratios of length to breadth) two classes of proteins are found:
- Globular proteins: They have an axial ratio of less than 10 (usually about 3 or 4), such as plasma albumins and globulins and many enzymes, and they have a spheroidal shape.
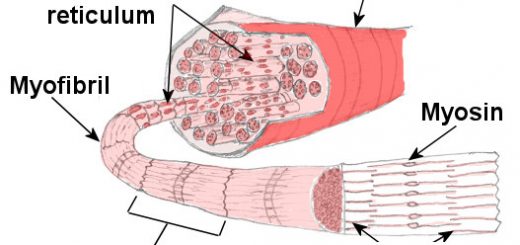
- Fibrous proteins: They have an axial ratio of more than 10, such as keratin, myosin fibrin, and collagen.
II. According to the biological value:
- Proteins of high biological value: i.e. they contain all the essential amino acids such as animal proteins.
- Proteins of low biological value: i.e. deficient in one or more essential amino acids e.g. plant proteins as Zein.
III. According to structure:
1- Simple proteins: Formed only by amino acids.
a- Protamines and histones are water-soluble basic proteins rich in histidine, arginine, and lysine, they are present in nucleoproteins (protamines are present in fish and histones are present in plants and animals.). Globin; the protein moiety of Hb and myoglobin is considered as histone.
b- Albumin (water-soluble) and globulins, (soluble in dilute salt solutions). These are heat coagulable, globular proteins of high biological value. Globulins have larger molecular weight compared to albumin, they are present mainly in blood plasma, egg white, and milk.
c- Scleroproteins (connective tissue proteins or albuminoids), these are fibrous structural proteins, they are insoluble in most protein solvent. They include:
i. Keratins (epidermal protein) is rich in sulphur containing amino acid (Cysteine), they are the proteins of the outer surface of the skin, hair, and nails. it is an α-helical polypeptide chain.
ii. Collagen is present mainly in the skin, cartilage, tendons, and ligaments, Collagens are the most important proteins in mammals, making up about 25% of the total proteins, there are many different types of collagen, particularly in connective tissue; they form insoluble tensile fibers that occur as structural elements of the extracellular matrix and connective tissue throughout the body.
Their name (which literally means “glue-producers”) is derived from the gelatins that appear as a decomposition product when collagen is boiled, collagen has an unusual amino acid composition. Approximately one-third of the amino acids are glycine (Gly). Collagen is largely present as a triple helix made up of three polypeptides (tropocollagen).
In each of three individual collagen chains (polypeptides) of tropocollagen, the triplet Gly-X-Y is constantly repeated in the sequence i.e., every third amino acid in such sequence is a glycine, with the X position often being occupied by Proline and the Y position by hydroxyproline also hydroxylysine occurs.
In triple helices, every third residue lies on the inside of the molecule, where there is only room for glycine residues (Glycine is the smallest amino acid), the two latter amino acids are only formed during collagen biosynthesis as a result of posttranslational modification, these hydroxylated amino acids are characteristic components of collagen.
The hydroxyproline residues stabilize the triple helix by forming hydrogen bonds between the chains, the strength of collagen comes from these H-bonding between chains. While the hydroxyl groups of hydroxylysine are partly glycosylated with a disaccharide (-Glc-Gal) and help linking tropocollagen molecules together. Numerous tropocollagen aggregate extracellularly into a defined arrangement forming cylindrical fibrils.
Nineteen different collagens are now known and they are distinguished using roman numerals, the various types of collagen consist of different combinations of chains. Types I, Il, and I represent 90% of collagens.
iii. Elastin: Found in yellow elastic tissue.
iv. Ossein: The main protein of bone and teeth.
2- Conjugated proteins (compound proteins): Those contain in addition to the protein moiety some other groups called the prosthetic group. According to the prosthetic group; they are classified into:
- Phosphoproteins: They contain phosphoric acid as a prosthetic group; conjugated to the hydroxyl group of serine or threonine. They are of animal origin e.g. Caseinogen of milk and vitellin of egg yolk. Caseinogen is converted by rennin to soluble casein which is precipitated by Ca as Ca caseinate (cheese).
- Glycoproteins: They are proteins that have carbohydrates (glycan) covalently attached to their polypeptide backbones. It is present in the mucus.
- Lipoproteins: These are combinations of proteins with lipids. They are present in cell membranes, and plasma lipoproteins.
- Nucleoprotiens: They contain nucleic acids (DNA or RNA) as prosthetic groups attached to protamines or histones, they are found in cell nuclei and also in the cytoplasm.
- Chromoproteins: These are proteins that contain colored prosthetic groups e.g. Haemoglobin is composed of globin and haem (red), also cytochromes, catalases, and peroxidases contain haem pigment conjugated to specific proteins. (myoglobin is similar to Hb but present in skeletal muscle).
- Metalloproteins: They contain metals as prosthetic groups, such as -Hb and ferritin contain iron, ceruloplasmin contains copper.
3- Derived proteins: They are hydrolytic products of proteins as a result of acids, alkalis of enzymes. Gelatin which is a hydrolytic product of collagen is considered as an example of derived protein, it is poor in essential amino acids, but it is used as a supplementary protein as it is easily digested.
Protein definition, structure, order, denaturation & Bonds responsible for protein structure
Protein structure, Classification, properties of amino acids & Formation of peptide bonds
Protein biosynthesis steps, site, importance, inhibitors & Protein maturation
DNA Repair types, definition & importance, Protein Biosynthesis & Genetic Code
Types of Derived lipids, Steroids, Animal sterols, Plant sterols & bile acids