Types of mixtures and solutions with examples, What’s the Difference?
Mixture means a thing that contains two or more different substances in any ratio such as seawater, granite, and gasoline, The mixtures can be classified according to their homogeneity into two types which are homogeneous (solutions) and heterogeneous (colloids and suspensions).
Types of mixtures and solutions
Mixtures and solutions are both combinations of substances, but they differ in how those substances are distributed and interact. In mixtures, the substances retain their individual properties, whereas, in solutions, the solute is uniformly dispersed within the solvent. Components of a mixture can be separated more easily than those of a solution.
Homogeneous
Solutions
Their components cannot be distinguished by the naked eye or the electron microscope, such as Table salt solution in water, Cane-sugar solution in water, and Cobalt (II) chloride solution in water.
Heterogeneous
Colloids
Their components can be distinguished by the electron microscope only, such as Aerosols, Hair gel, Mayonnaise emulsion, Blood, and Milk.
Suspensions
Their components can be distinguished by the naked eye, Such as Table salt in kerosene, cane sugar in kerosene, Cobalt (II) chloride in kerosene, and oil in water.
Homogeneous mixtures
Solutions
Solutions are necessary for the biological processes that occur inside living organisms, If you add a small quantity of a substance (such as sugar) to a large amount of another substance (such as water) and disappear in it, Therefore, Sugar is a solute, water is a solvent and the produced mixture is a solution.
The solute is the minor component that has the lesser ratio in the solution, the solvent is the major component that has the larger ratio in the solution, A solution is a homogeneous mixture of two or more substances that are chemically unreacted.
The sweet taste of the sugar solution in water is the same in all of its parts because of the homogeneity of the sugar solution, as each part of the solution contains the same amount concentration of sugar.
Types of solutions
Solutions can be classified according to the physical state of the solvent, The ability to conduct electricity, and the degree of saturation.
According to the physical state of the solvent
Solutions are divided into gaseous solutions, liquid solutions, and solid solutions.
- Gaseous solutions: when the solute is gas and solvent is gas such as atmospheric air and natural gas, when the solute is gas and the solvent is a liquid such as soft drinks & oxygen dissolved in the water.
- Liquid solutions: when the solute is liquid and the solvent is liquid such as alcohol in water, ethylene glycol (antifreeze) in water, when the solute is solid and the solvent is liquid such as sugar or salt in water.
- Solid solutions: when the solute is gas and the solvent is solid such as hydrogen gas on palladium or platinum, when the solute is liquid and the solvent is solid such as liquid mercury dissolved in solid silver ( silver amalgam ) Hg (l) / Ag (s), when the solute is solid and the solvent is solid such as alloys (nickel-chrome alloy).
Scientific background knowledge
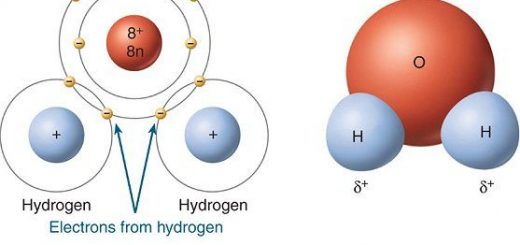
Electronegativity is the ability of an atom to attract the electrons of the bond to itself, A polar bond is a covalent bond between two atoms that are different in electronegativity, The higher electronegative atom carries a partial negative charge ( δ− ), while the other atom carries a partial positive charge (δ+).
Polar molecules are molecules that have an end carrying a partial positive (δ+) and another end carrying a partial negative charge (δ−), A Polar compound is a compound that has a polar covalent bond, The degree of polarity depends on the difference in electronegativity between the bonded atoms, Geometry of the molecule and bond angles.
Water is considered as a famous polar solvent because this polarity is attributed to the presence of two polar bonds (because the electronegativity of oxygen is higher than that of hydrogen), The structure of the water molecule contains a large bond angle = 104.5 degrees.
Types of Mixtures
There are two main types of mixtures, They are categorized based on how the components are distributed: heterogeneous mixtures and homogeneous mixtures.
- Heterogeneous Mixture: The components are not uniformly distributed, and the different parts can usually be seen. Example: Salad, sand, and water.
- Homogeneous Mixture: The components are uniformly distributed, and the mixture looks the same throughout. Example: Salt and sugar mixed.
1. Heterogeneous Mixtures
A heterogeneous Mixture is a mixture in which the components are not uniformly distributed, The different parts of the mixture can often be seen and separated physically, The individual components remain distinct and are visible. Examples of separation methods: are filtration, decantation, or sieving.
Examples:
- Sand and water: The sand settles at the bottom while the water stays on top.
- Trail mix: Different nuts, raisins, and candies can be separated.
2. Homogeneous Mixtures
A homogeneous mixture is a mixture in which the components are distributed, and the composition is uniform throughout. The mixture looks the same throughout, and the individual components are not visible. The composition is consistent, meaning every part of the mixture has the same proportions of components.
Examples:
- Saltwater: Salt dissolves in water, forming a uniform solution.
- Air is a mixture of gases such as nitrogen, oxygen, and carbon dioxide that are distributed.
- Alloys such as Brass (a mixture of copper and zinc) and steel (a mixture of iron and carbon).
Other Types of Mixtures
- Solid-Solid Mixtures: All the components are in a solid state (e.g., alloys, such as bronze or brass).
- Solid-Liquid Mixtures: Solids dispersed in liquids (e.g., muddy water).
- Liquid-Liquid Mixtures: Two liquids mixed together (e.g., vinegar and water).
- Gas-gas mixtures: Gases mixed together (e.g., air).
- Gas-Liquid Mixtures: A gas dissolved in a liquid (e.g., carbonated drinks).
You can follow science online on YouTube from this link: Science online
You can download Science Online application on Google Play from this link: Science online Apps on Google Play
Solutions of electrolytes & non-electrolytes and Degree of saturation
Types of mixtures in terms of homogeneity
Matter, Mixtures Types, Properties, Formation and Separation