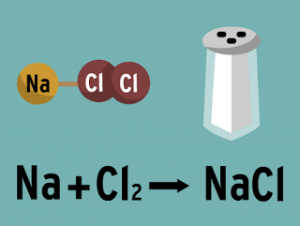Salts formation methods & types of aqueous solutions of salts
Salts formation methods
Salts can be practically prepared by Reaction of active metals with diluted acids , Reaction of metal oxides with acids , Reaction of metal hydroxides with acids and Reaction of metal carbonates or bicarbonates with some acids .
Reaction of active metals with diluted acids
Active metal + acid ( dil ) → Salt of acid + Hydrogen gas ↑
The metal above hydrogen in the chemical activity series replace hydrogen in the diluted acid solutions, giving salt of acid and hydrogen gas is evolved which burns with a pop sound.
The salt of acid which is produced dissolves in the water and can be separated by heating the solution, where the water evaporates and the salt remains.
Example : Reaction of zinc with diluted sulphuric acid .
Zn ( s ) + H2SO4 ( aq ) → ZnSO4 ( aq ) + H2 (g) ↑
Reaction of metal oxides with acids
Metal oxide + Acid ( dil ) → Metal oxide + Acid ( dil )
This method is used if there is a difficulty in the direct reaction of metal with acid because the reaction is dangerous ( e.g. the reaction of sodium with hydrochloric acid is vigorous), The metal doesn’t react with the acid , because it is less reactive than hydrogen ( e.g. copper doesn’t react with hydrochloric acid ) .
Example : Reaction of copper oxide with sulphuric acid .
CuO( s ) + H2SO4 ( aq ) → CuSO4 ( aq ) + H2O (l) ↑
Reaction of metal hydroxides with acids
This method is suitable in the case of metal hydroxides , that are dissolving in the water and considered as alkalis, These types of reactions are known as Neutralization reactions .
Neutralization reactions can be used in analytical chemistry to calculate the concentration of an acid ( or an alkali ) using a known concentration of a base ( or an acid ) in the presence of a suitable indicator.
Neutralization point ( End point ) is the moment at which the amount of acid is equivalent to the amount of alkali .
Metal hydroxide ( alkali ) + Acid ( dil ) → Salt of acid + Water
NaOH ( aq ) + HCl ( aq ) → NaCl ( aq ) + H2O ( l )
Reaction of metal carbonates or bicarbonates with some acids
Metal carbonates ( or bicarbonates ) + Acid → Salt of acid + Water + Carbon dioxide gas ↑
Metal carbonates or bicarbonates are salts of carbonic acid which is an unstable acid , due to its low boiling point .
It is possible for any other acid ( that is more stable than it ) to remove it from its salt solutions to form a new acid salt.
Carbonic acid decomposes into water and carbon dioxide gas , which turns the clear limewater into milky.
Example : Reaction of sodium carbonate with hydrochloric acid
Na2CO3 ( s ) + 2 HCl ( aq ) → 2NaCl ( aq ) + H2O ( l ) + CO2 ( g ) ↑
The reaction of metal carbonates or bicarbonates with some acids is called acidity test because this reaction is used to detect the presence of acids , as a strong effervescence occurs and CO2 gas evolves, which turns the clear limewater into turbid.
Types of aqueous solutions of salts
The aqueous solution of sodium chloride is neutral because NaCl is produced from the reaction between a strong acid ( HCl ) and a strong base ( NaOH ) , So , the pH value is 7 .
The aqueous solution of sodium carbonate is basic because Na2CO3 is produced from the reaction between a strong base ( NaOH ) and a weak acid ( H2CO3 ) , So , its pH value > 7 .
The pH value of an aqueous solution of ammonium chloride is less than 7 because this solution is an acidic , where NH4Cl is produced from the reaction between a strong acid ( HCl ) and a weak base ( NH4OH ) .
The pH value of an aqueous solution of ammonium acetate equals 7 because this solution is a neutral, where CH3COONH4 is produced from the reaction between a weak acid ( CH3COOH ) and a weak base ( NH4OH ).
Types of compounds, Properties of Acids, Bases (alkalis), Oxides and Salts
Salts formation, The chemical formula of salts and its naming
Economic importance of some common acids, bases and salts (minerals)