States of water, physical and chemical properties of the water, What is the water cycle?
Physical properties of the water
The water has a higher melting point, boiling point, and heat of vaporization than most common liquids. This fact indicates that there are strong forces of attraction between the adjacent water molecules.
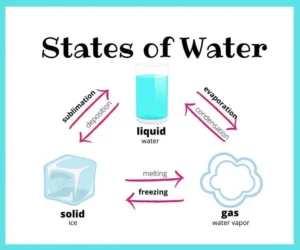
Water exists in three states
| States of water |
The states of water are the solid-state (ice), the liquid state (water), and the gaseous state (water vapour) at normal temperature.
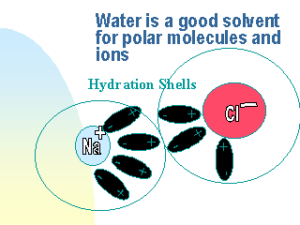
The water is a good polar solvent
You notice that the water is a good polar solvent. So, it has a great ability to dissolve the most ionic compounds, such as table salt (sodium chloride).
The water can also dissolve some covalent compounds, such as sugar, which can form hydrogen bonds with it. Some covalent compounds, such as oil, can not dissolve in water as they can not form hydrogen bonds with water.
 |
| The water is a good polar solvent |
Pure water boils at 100 degrees Celsius and freezes at 0 degrees Celsius. The high boiling point and the low freezing point of water are due to the presence of the hydrogen bonds between its molecules.
The water density decreases on freezing
The density of the water in the solid-state (the ice) is lower than its density in the liquid state when the temperature of the water decreases by 4 degrees Celsius.
The water molecules are collected together by the hydrogen bonds to form the ice crystals, which have a hexagonal shape, a large volume, and a large number of spaces between them.
The ice crystals float on the water surface, and this helps in the preservation of the life of the aquatic creatures in it.
The water has a high latent heat
The water has high latent heat. So, it resists the change from one state to another. So, the water is used to extinguish the fires, as it consumes a large amount of the heat of the combustion media during its vaporization process.
The water has a high specific heat
You notice that the temperature of the human body does not change when the atmospheric temperature changes. The high specific heat of the water makes it absorb or lose a large amount of heat without changing its temperature.
Chemical properties of the water
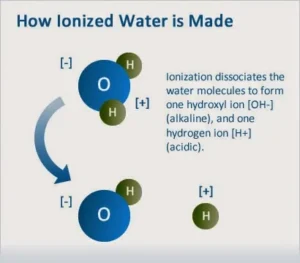
The weakness of the water ionization
| The water ionizes to give positive hydrogen ions and negative hydroxide ions |
The ionization is the process of converting the molecules of some covalent compounds into ions, and pure water is considered weakly ionized, which gives positive hydrogen ions and negative hydroxide ions.
The water has a neutral effect
The water has a neutral effect on the litmus paper, as when it is ionized, it gives equal numbers of the positive hydrogen ions (which are responsible for the acidic property) and the negative ions ( which are responsible for the basic property).
The resistance of the water to decomposition
The water does not decompose into its elements (hydrogen and oxygen) under normal conditions or even by the effect of the heat that helps to keep the aqueous solutions inside the cells of the living organisms.
FAQ about Water
Water is one of the most essential substances on Earth. It exists in different states and has unique physical and chemical properties that make it vital for life and many natural processes. The following are frequently asked questions about the states of water and its physical and chemical properties.
1. What are the states of water?
Water exists in three main states:
- Solid (Ice): Water freezes into ice at 0°C.
- Liquid (Water): The most common state found in rivers, lakes, and oceans.
- Gas (Water vapor): When water evaporates or boils, it becomes vapor in the atmosphere.
2. What is the water cycle?
The water cycle is the continuous movement of water on Earth through processes such as:
- Evaporation.
- Condensation.
- Precipitation.
- Collection in rivers, lakes, and oceans.
This cycle helps maintain the balance of water in nature.
3. What are the physical properties of water?
Physical properties of water are characteristics that can be observed without changing its chemical composition. These include:
- Water is colorless, odorless, and tasteless.
- It has a boiling point of 100°C at normal atmospheric pressure.
- It freezes at 0°C.
- It has high surface tension.
- It has high heat capacity, meaning it can absorb a lot of heat before its temperature changes.
4. Why does water have high surface tension?
Water molecules are strongly attracted to each other through hydrogen bonds. These bonds create strong cohesion between molecules, resulting in high surface tension.
5. What are the chemical properties of water?
Chemical properties describe how water reacts with other substances. Important chemical properties include:
- Water can act as a solvent, dissolving many substances.
- It participates in chemical reactions, such as hydrolysis.
- Water can react with metals to form hydroxides and hydrogen gas.
- It is involved in biological processes like photosynthesis and respiration.
6. Why is water called the universal solvent?
Water is called the universal solvent because it can dissolve more substances than most other liquids. This ability helps transport nutrients and minerals in living organisms and the environment.
7. What happens when water freezes?
When water freezes:
- Molecules slow down.
- Hydrogen bonds arrange molecules into a crystal structure.
- The volume increases, making ice less dense than liquid water.
This is why ice floats on water.
8. What happens when water boils?
- When water reaches its boiling point:
- Molecules gain enough energy to escape into the air.
- Liquid water changes into water vapor (gas).
9. What is cohesion and adhesion in water?
- Cohesion: Attraction between water molecules.
- Adhesion: Attraction between water molecules and other surfaces.
These properties help water move through plant stems and soil.
10. Why is water important for living organisms?
Water is essential because it:
- Supports cellular functions.
- Helps transport nutrients and waste.
- Regulates body temperature.
- Participates in chemical reactions in cells.
11. How do the properties of water affect the environment?
Water’s unique properties help regulate Earth’s climate, support ecosystems, and maintain stable temperatures in oceans and lakes.
You can subscribe to Science Online on YouTube from this link: Science Online
Water properties, structure, pollutants, & protection of water from pollution
The importance of the water and its structure
Ionization of water, Hydrogen Exponent (pH value), Solubility product & Hydrolysis of salt solutions
The water pollutants and the protection of the water from pollution