Properties of Metals during chemical reactions and reaction of active metals, Why are active metals dangerous?
Metals are elements that are typically: Shiny (lustrous), Good conductors of heat and electricity, Malleable (can be hammered into sheets), Ductile (can be drawn into wires), and Solid at room temperature (except mercury).
Metals
Metals are the elements that have less than four electrons in their outermost energy levels. During chemical reactions, the atoms of the metallic elements tend to lose their outermost electrons and change into positive ions.
The positive ions carry a number of positive charges equal to the number of lost electrons. The electronic structure of the positive ion is similar to that of the nearest preceding inert gas in the modern periodic table.
Some metals, such as Magnesium (Mg) and iron (Fe), react with oxygen, giving the metals oxides, which are called “Basic oxides”. Basic oxides are metallic oxides, and some of them dissolve in water, forming alkaline solutions.
Some metal oxides (such as magnesium oxides) dissolve in the water, giving alkalis (metal hydroxide) which turn the litmus solution into blue. Some metal oxides, such as iron II oxide, do not dissolve in water. All alkalis are bases, while not all bases are alkalis.
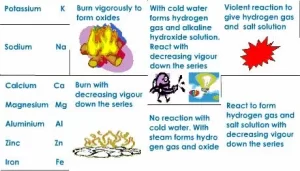
The reaction of the metals with the water depends on the position of the metal in the chemical activity series. The chemical activity series is a series in which metals are arranged in descending order according to their chemical activity.
Potassium and Sodium react instantly with water, and hydrogen gas evolves, which burns with a pop sound. Calcium and Magnesium react very slowly with cold water. Zinc and Iron react with hot water vapour at high temperatures only. Copper and Silver do not react with water.
Some active metals, such as magnesium (Mg) and zinc (Zn), react with dilute acids (such as hydrochloric acid), giving the salt of the acid and hydrogen gas evolved. The inactive metals, such as copper (Cu), do not react with dilute acids, and no gas evolves.
What are Active Metals?
Active metals are metals that are highly reactive, especially with water, oxygen, and acids. They tend to lose electrons easily to form positive ions (cations), making them quick to take part in chemical reactions.
Examples of Active Metals
These are mainly found in Group 1 (alkali metals) and Group 2 (alkaline earth metals) of the periodic table:
Group 1: Alkali Metals
- Lithium – Li – High Reactivity.
- Sodium- Na – Very High Reactivity.
- Potassium – K – Extremely High Reactivity.
- Rubidium – Rb – Super reactive.
- Cesium – Cs – Extremely reactive.
Properties:
- React violently with water.
- Form strong bases (alkaline solutions).
- Soft and shiny (but tarnish quickly in the air).
- Stored in oil to prevent reaction with moisture/oxygen.
Group 2: Alkaline Earth Metals
- Calcium – Ca – Reactive.
- Strontium – Sr – Very Reactive.
- Barium – Ba – Very Reactive.
Properties:
- React with water (less violently than Group 1).
- Form oxides and hydroxides.
- Also, form alkaline solutions.
Reactions of Active Metals
- With Water: Produces metal hydroxide and hydrogen gas. Example: 2K + 2H2O→ 2KOH + H2↑
- With Oxygen: Forms metal oxides. Example: 4Li + O2 → 2 Li2O
- With Acids: Produces salt and hydrogen gas. Example: Ca + 2HCl → CaCl2 + H2↑
Reactions of active metals with water or acid are exothermic (release heat). Some (like Na and K) can ignite or explode during the reaction. Proper lab safety is essential when handling.
Properties of Metals During Chemical Reactions
Metals tend to lose Electrons (Oxidation). Metals are generally electropositive, meaning they easily lose electrons to form positive ions (cations). Example: Na → Na + e−
Form Ionic Compounds: Metals usually react with non-metals (like oxygen, chlorine, sulfur) to form ionic compounds. Example: Mg + Cl2 → MgCl2
React with Oxygen to Form Oxides: Most metals react with oxygen to form metal oxides, which are basic in nature. Example: 4K + O2 → 2 K2O
Displace Less Reactive Metals from Solutions: More reactive metals can displace less reactive ones from their salt solutions (based on the reactivity series). Example: Zn + CuSO4 → ZnSO4 + Cu
Reaction of Active Metals
Active metals (like potassium, sodium, calcium, etc.) react very vigorously—sometimes even violently—with:
- Water: Highly reactive metals (Group 1 and 2) react with water to form metal hydroxide and hydrogen gas. This reaction releases a lot of heat; sodium and potassium can catch fire.
- Acids: Active metals also react with dilute acids like HCl or H₂SO₄, producing salt and hydrogen gas.
- Air/OxygenL They react quickly with oxygen, forming oxides. Some even need to be stored in oil to prevent reaction with air (like Na, K).
FAQ about Properties of Metals
Properties of Metals During Chemical Reactions
1. What happens to metals during chemical reactions?
Metals usually lose electrons and form positive ions (cations).
2. Why do metals lose electrons easily?
Because they have low ionization energy and a weak attraction between the nucleus and outer electrons.
3. What type of compounds do metals form?
Metals mainly form ionic compounds when they react with non-metals.
4. How do metals react with oxygen?
Metals react with oxygen to form metal oxides, which are usually basic.
Example: Iron + Oxygen → Iron oxide (rust)
5. How do metals react with water?
Highly active metals (like sodium) react violently with water. Less active metals react slowly or not at all
Example: Sodium + Water → Sodium hydroxide + Hydrogen gas
6. How do metals react with acids?
Metals react with acids to produce:
➡️ Salt + Hydrogen gas
Example: Zinc + Hydrochloric acid → Zinc chloride + Hydrogen
7. What happens when metals react with chlorine?
They form metal chlorides (ionic compounds).
Reactions of Active Metals
8. What are active metals?
Active metals are metals that react quickly and easily with other substances.
Examples: Potassium, Sodium, Calcium
9. What is the reactivity series?
It is a list that arranges metals based on how reactive they are (from most to least reactive).
10. How do active metals react with water?
Produce metal hydroxide + hydrogen gas
Reaction is often fast and exothermic
11. Why are active metals stored in oil?
To prevent reaction with air and moisture, which can be dangerous.
12. How do active metals react with acids?
They react rapidly, producing large amounts of hydrogen gas.
13. What is a displacement reaction?
A more reactive metal can displace a less reactive metal from its compound.
Example: Iron + Copper sulfate → Iron sulfate + Copper
14. Why are active metals dangerous?
Because they can react violently with water or air, sometimes causing explosions.
15. Do all metals react the same way?
No, their reactivity depends on their position in the reactivity series.
16. What is the role of hydrogen in these reactions?
Hydrogen is released as a gas when metals react with acids or water.
Metals tend to lose electrons in reactions. Active metals react quickly and strongly. Common reactions include oxygen, water, and acids. Reactivity determines how metals behave
You can follow Science Online on YouTube from this link: Science Online
Metallic & nonmetallic properties, Acidic & basic properties in the periodic table
Modern periodic table and classification of Elements
Radius property, Ionization potential, Electron affinity and Electronegativity
Chemical combination, Properties of Metals, Nonmetals and Noble (inert) gases
Elements of s-block, Properties of the first group elements 1A (Alkali metals) in the periodic table