How can you calculate the valency of each element?
The valency
The valency is the number of electrons that an atom gains, loses or even shares during a chemical reaction, The valency is the number of electrons that an atom needs to gain or lose in order to achieve noble gas electronic configuration.
In the chemical reaction, the atoms try to reach the stable state by losing their outermost electrons (as in case of the metals) or by sharing a number of electrons with other atoms (as in case of nonmetals).
The valency of the elements or the ions to write the chemical formula of a compound, Chlorine has a valency of 1 as it needs to gain 1 electron to achieve noble gas electronic configuration.
The atoms try to reach the stable state by gaining a number of electrons to complete the outer shell by (8) electrons (as in the case of nonmetals).
The outermost electrons of an atom determine its valency, the valency of an element measures its ability to combine with the other elements, The valency is determined by the number of electrons in the outer shell of each atom of an element.
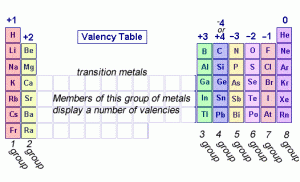
The valency of some elements in the modern periodic table
The elements in the group (1 A) in the modern periodic table have one valent electron in their outer shell, they are very active, they are mono-valent elements as they have only one electron in their outermost energy levels.
The valency of the alkaline earth metals in the group (2 A) is divalent, they are divalent elements as they have two electrons in their outermost energy levels.
The elements of the group (7 A) are mono-valent elements as their outermost energy levels have (7) electrons, The elements in the group (18) in the modern periodic table are the noble gases which have a valence of zero as the valence shell is saturated with 8 electrons.
To determine the valency of the ionic molecule phosphorus tetraoxide (PO4, four atoms of oxygen and one atom phosphorus) you should multiply the total valency of the four oxygen atoms (valency 2) and subtract that from the valency of the phosphorus atom (valency 5), That reveals the valency of (PO4) is 3.
The valency of an element is always a whole number, and some elements exhibit more than one valency, they have more than one valency such as sulphur (S) is divalent, tetravalent and hexavalent.
Nitrogen (N) is trivalent and pentavalent while phosphorus (P) is trivalent and pentavalent, The valency of copper I is monovalent while the valency of copper II is divalent.
The valency of Iron [II] sulfate or ferrous sulfate is divalent as in FeSO4, the valency of iron is two while the valency of Iron [III] sulfate or ferric sulfate is trivalent as in Fe2(SO4)3, the valency of iron is three.
What is Valency? Valencies of some metallic, nonmetallic elements & atomic groups
Modern periodic table and classification of Elements
The quantum numbers and principles of distributing electrons
Metallic & nonmetallic property, Acidic & basic property in the periodic table
Theories explaining the covalent bond, Octet rule & Overlapped orbitals concept




It helped me a lot i can now understand what valency is
Thank you very much for your comment
it helped me a lot…………you are doing very good for students keep it up…………..thanks a lot!!!
You are welcome
Thank you very much for your comment
thank you so much.. i can clearly understand about valency now…
You are welcome
Thank you very much for your comment
This helped to understand the valency.good efforts.
Thank you very much for your comment
its help very much
thanking you heartfully
You are welcome
Thank you very much for your comment
P+O2 =P2O3O
Number of atoms of reactants must equal number of atoms of products
4P + 3O2 = 2P2O3
How valency and reaction done.
4P + 3O2 = 2P2O3
Number of atoms of reactants must equal number of atoms of products
Can you explain me haber process
You can read this article about Preparation of ammonia gas in industry ( Haber-Bosch’s method )
The most famous elements in 5A group ( Nitrogen properties , Preparation & compounds )
baie dankie
This article really helped me …thanks
Cover more and more topics under your articles ..
You are welcome