Functions of Kidneys, Role of Kidney in glucose homeostasis, Lipid and protein metabolism
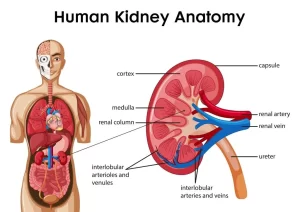
The kidneys are two bean-shaped organs, each about the size of a fist, They are located below the rib cage, one on each side of your spine, The kidneys are powerful chemical factories that remove waste products from the body, they remove drugs from the body, they can balance the body’s fluids, they can release hormones that regulate blood pressure, and they can control the production of red blood cells.
Functions of the kidney
I. Homeostatic function
The kidney maintains the constancy of the internal environment through the excretion of urine, Urine contains excess water, electrolytes, acids, and alkalis, Moreover, it contains the waste products of metabolism, so, the kidney plays an important role in:
- Maintenance of water balance.
- Maintenance of electrolyte balance.
- Maintenance of blood pH.
II. Endocrine function
- The juxtaglomerular cells of the kidney secrete renin which leads to the formation of angiotensin II which regulates arterial blood pressure.
- The JG cells secrete erythropoietin hormone that stimulates the bone marrow to increase the regeneration of red blood cells.
- The kidney converts vitamin D3 to an active form 1,25-dihydroxycholecalciferol which promotes the absorption of calcium from the intestine.
- Moreover, the kidney secretes prostaglandins.
III. Metabolic function of the kidney
The energy supply of the kidney
The kidney tissue represents less than 0.5 % of the body weight; it receives 25 % of the cardiac output and 10 % of O2 consumption, this is required for the synthesis of ATP needed to reabsorb most of the solutes filtered through the glomerular membranes. The energy reserves in the kidney (glycogen, phosphocreatine, or neutral lipids) are very low, thus the kidney must get its energy requirement from circulating substrates.
Substrates used by the kidney for energy production: In the fed state, the kidney mainly oxidizes lactate (33% of total renal O2 consumption) and glucose. While during starvation, there is a decrease in blood lactate and glucose concentrations and an increase in blood ketone bodies and fatty acids concentrations, so the renal uptake of ketone bodies and fatty acids increases, they appear to be the major kidney fuel during starvation.
A) Carbohydrate metabolism in the kidney
The following metabolic pathways occur in the kidney: Glycolysis, Citric acid cycle, HMS, Fructose metabolism, and gluconeogenesis.
Role of the kidney in glucose homeostasis
Regarding glucose homeostasis, the kidney can be considered as 2 organs:
- Renal medulla: in which glucose utilization occurs mainly.
- Renal cortex: in which glucose synthesis (gluconeogenesis) occurs mainly.
This occurs due to the differences in the distribution of various enzymes in the renal medulla and renal cortex, In the renal medulla, the cells have considerable glucose-phosphorylating enzymes (hexokinase), glycolytic enzyme activity and depend on glucose as a major source of energy.
The cells don’t have glucose-6-phosphatase and other gluconeogenic enzymes, Thus cells of the adrenal medulla can take up, phosphorylate and metabolize glucose through glycolysis; they can form glycogen, but cannot release free glucose into the circulation.
In the renal cortex, the cells have gluconeogenic enzymes, have little phosphorylating enzymes, and cannot synthesize appreciable concentrations of glycogen under normal conditions, So, we can say that the release of glucose by the normal kidney is exclusively, a result of renal cortical gluconeogenesis, The most important substrates for renal gluconeogenesis are glutamine and lactate, followed by glycerol, while alanine is preferentially used by the liver.
Renal glucose metabolism in the postprandial conditions
After meal ingestion, glucose is mainly utilized by the brain, liver & muscles, There is a decrease in liver gluconeogenesis. Renal glucose uptake is < 10% of the ingested glucose, Renal gluconeogenesis paradoxically increases and it accounts for 50% of the endogenous glucose release, This may take place due to:
- Increase in the availability of gluconeogenic precursors (e.g. lactate and amino acids).
- This increase in renal gluconeogenesis helps liver glycogen repletion by suppression of hepatic glucose release.
Hormonal control of renal gluconeogenesis
Epinephrine: it has more effect on renal gluconeogenesis than hepatic gluconeogenesis (may be due to the rich autonomic innervation of the kidney). The mechanism is mediated through free fatty acids released during lipolysis accompanying hypoglycemia.
After prolonged fasting
In humans, the early increase in glucose release is mainly caused by hepatic glycogenolysis, later it is mainly a result of gluconeogenesis, Renal glucose uptake is reduced, and renal glucose release increases, Hepatic glucose release decreases by 25%, thus, the liver cannot compensate for the kidney to preserve normoglycemia in patients with renal insufficiency during prolonged fasting, This may explain why patients with renal failure develop hypoglycemia.
B) Lipid metabolism in the kidney
The kidney has an active glycerol kinase enzyme, It is a site for:
- B-Oxidation.
- Synthesis of carnitine.
- ketolysis.
- Denovo synthesis of cholesterol.
- Denovo synthesis of fatty acids.
C) Protein metabolism in the kidney
The kidney is a site for:
- Urea synthesis.
- Oxidative deamination and trans-deamination of individual amino acids.
- Creatine formation from glycine and arginine.
- It is a major site for the action of glutaminase enzyme with its great role in acidosis.
D) Other Metabolic Functions of the Kidney
Ammonia production in the kidney: Ammonia is produced in the renal tubules by the action of the enzyme glutaminase and L- glutamate dehydrogenase. lt combines with H+ to form ammonium ions: NH3+↔ H+ NH4+.
This reaction is favored at the acid pH of urine, The formed NH4+ in the tubular lumen cannot easily cross the cell membranes and is trapped in the lumen to be excreted in urine with other anions such as phosphate, chloride, and sulphate. NH4+ production in the tubular lumen accounts for about 60 % excretion of hydrogen ions associated with nonvolatile acids, In renal insufficiency, the kidneys are unable to produce enough NH3 to buffer the nonvolatile acids leading to systemic acidosis.
You can subscribe to Science online on YouTube from this link: Science Online
You can download Science Online application on Google Play from this link: Science Online Apps on Google Play
Urinary system structure, function, anatomy, organs, Blood supply and Importance of renal fascia
Suprarenal glands (adrenal glands) anatomy, structure and Effect of Deficiency of Catecholamines
Histological structure of kidneys, Uriniferous tubules and Types of nephrons
Urine formation, Factors affecting Glomerular filtration rate, Tubular reabsorption and secretion
Functions of the loop of Henle, distal tubules, collecting ducts and Glucose tubular maximum