Serine function, Tyrosine, tryptophan, Pellagra (Vitamin B3 Deficiency) and Nucleotide metabolism
Serine is used in the biosynthesis of proteins, It is an α-amino acid, Serine is classified as a nutritionally nonessential (dispensable) amino acid, but metabolically, serine plays an essential role in many cellular processes, It is the main source of one-carbon units for methylation reactions that occur via the generation of S-adenosylmethionine.
Serine
It is a non-essential hydroxy-containing and glucogenic amino acid.
Synthesis of serine
- From glycolytic intermediates (3-phosphoglycerate).
- From glycine by serine hydroxymethyl transferase reaction.
Catabolism of serine
- Conversion to glycine, then by glycine cleavage system, it is cleaved to CO2 & NH3.
- Conversion to pyruvate by serine dehydratase (serine is a glucogenic amino acid).
Functions and derivatives
- It provides the carbon skeleton of cysteine.
- It forms glycine by serine hydroxymethyltransferase.
- Synthesis of ceramide (conjugation with palmitoyl CoA).
- Synthesis of ethanolamine by decarboxylation. Thus it is essential for the synthesis of phospholipids and sphingomyelin. (It is a lipotropic factor).
- It is a source of one carbon moiety.
- Covalent modification of enzyme activity by addition or removal of P as in glycogen synthase, glycogen phosphorylase and hormone-sensitive lipase.
Phenylalanine / Tyrosine
They are aromatic amino acids, Phenylalanine is an essential amino acid, while tyrosine is not essential in the presence of a good supply of phenylalanine, Hydroxylation of Phenylalanine gives rise to tyrosine.
I- Synthesis of Tyrosine
Il- Catabolic pathways of phenylalanine
There are 2 pathways for the catabolism of phenylalanine:
- Direct pathway (minor) where phenylalanine transamination reaction forms phenyl pyruvic acid which is excreted in urine via its metabolites, It is an alternative pathway of phenylalanine catabolism in phenylketonuria.
- Phenylalanine is transformed to tyrosine (major pathway) in the liver then tyrosine is catabolized to fumaric acid and acetoacetic acid i.e. phenylalanine and tyrosine are both glucogenic and ketogenic.
Ill- Functions and derivatives
Tyrosine forms special important compounds such as:
1. Melanin: in the melanocytes (pigment cells) in the skin, hair, and eye.
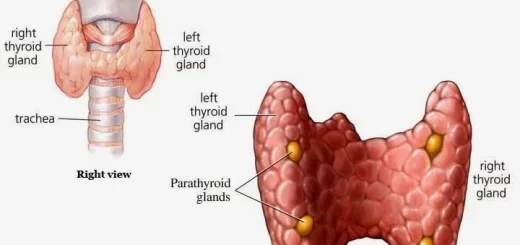
2. Tyrosine forms thyroid hormones, This occurs in the thyroid gland. Tyrosine is iodinated to monoiodotyrosine (3 MIT) and diiodotyrosine (3,5 DIT) then by coupling:
- Monoiodotyrosine + Di-iodotyrosine gives T3 (Triiodothyronine).
- Di- lodotyrosine + Di-iodotyrosine gives T4 (Thyroxine).
Both reactions occur in the thyroglobulin, Then T3 and T4 are released.
3) Tyrosine forms catecholamines (dopamine (1), norepinephrine (2), and epinephrine (3)). This occurs in cells of neural origin and in the adrenal medulla.
One of the principal metabolites of epinephrine and norepinephrine is vanillyl mandelic acid (VMA) which increases in the urine of patients with pheochromocytoma (tumour of the adrenal medulla).
IV- Metabolic Disorders of phenylalanine and Tyrosine catabolism
1- Phenylketonuria (PKU)
It is an inherited metabolic disorder of phenylalanine caused by defective liver phenylalanine hydroxylase or dihydrobiopterin reductase. The disease is characterized by increased phenylacetic acid, phenyl acetyl glutamine, phenyl pyruvic, and phenylacetic acid in the blood and urine.
The signs and symptoms include:
- Mental retardation.
- Eczema of the skin.
- The mousy odour of urine.
- Hypopigmentation as a result of decreased melanin pigment will lead to fair skin and hair and blue eyes.
The disease could be diagnosed by increased plasma phenylalanine levels (>20 mg/dl) (normal: 0.7 – 4 mg/dl); and by using a ferric chloride test which gives a blue-green colour with urine, Treatment is through a diet of low in phenylalanine and rich in tyrosine.
2- Tyrosinemia or tyrosinosis:
Inherited metabolic disorders are characterized by high levels of plasma tyrosine, Death usually occurs early from liver failure, Treatment is by low tyrosine and phenylalanine diet, The most important types are:
- Hepatorenal type (Type I tyrosinemia) due to fumaryl acetoacetate hydrolase deficiency.
- Oculocutaneous type (Type II tyrosinemia) due to hepatic tyrosine transaminase deficiency.
- Neonatal tyrosinemia due to defective p-hydroxy phenyl pyruvate hydroxylase.
3- Alkaptonuria
Inherited metabolic disorder due to defect in homogentisate oxidase, This causes the accumulation of homogentisate in blood and urine, Homogentisate is oxidized into a brownish-black pigment that causes the darkening of urine on standing in air, There is also arthritis and generalized pigmentation of connective tissue (Ochronosis))
4- Albinism
Hypomelanosis (decreased pigmentation) is due to defects in the eye, hair, and skin melanocyte. The most severe is due to a deficiency of tyrosinase enzyme in the skin, hair, and eyes. So, melanin pigments will not be formed leading to the white colour of the skin and making it sensitive to light which may lead to burning and carcinoma. Lack of pigments in hair causes fair hair, and lack of pigments in the eyes causes photophobia. The patient is called Albino.
Tryptophan
It is an essential, glucogenic, ketogenic, and heterocyclic amino acid.
I- Catabolic pathway
Tryptophan is catabolized through the kynurenine-anthranilate pathway to acetoacetyl COA (ketogenic) and alanine is developed through this pathway (glucogenic). Also, niacin can be synthesized through this pathway.
Catabolic pathway of tryptophan
Niacin is one of the B vitamins and enters in the formation of NAD and NADP 60 mg. Tryptophan can form 1mg of niacin. Its deficiency causes pellagra.
Pellagra
Pellagra is a disease caused by severe niacin deficiency. It is characterized by:-
1- Disturbance in lipid, carbohydrate, and protein metabolism.
2- 3 D’s:
- Diarrhea, alternating with constipation.
- Dermatitis in the sun-exposed areas.
- Dementia or progressive loss of all cerebral functions.
Causes of Pellagra
- Diet poor in both niacin and tryptophan e.g. eating maize as the sole diet which contains a protein called zein that is low in tryptophan.
- Malignant carcinoid syndrome in which tryptophan metabolism is directed into the formation of serotonin.
- Hartnup disease in which there is impairment of tryptophan absorption.
- Vitamin B6 deficiency potentiates niacin deficiency, as in the isoniazid drug, used in the treatment of tuberculosis.
II- Functions and derivatives (conversion to specialized products)
Synthesis of:
1- Niacin (through kynurenine-anthranilate pathway)
2- Conversion to serotonin (Hormone):
1% of Trp is normally converted to serotonin. This is done by hydroxylation of tryptophan by tryptophan hydroxylase in the liver. This is a mixed-function oxygenase that forms 5 hydroxytryptophans then it is decarboxylated to serotonin (5 hydroxy tryptamine) by pyridoxal phosphate enzyme. Serotonin is found in the intestine, platelets, and central nervous system.
In argentaffinoma (malignant carcinoid syndrome) which is serotonin producing. tumor cells in the argentaffin tissue of the abdominal cavity, there is overproduction of serotonin, where up to 60% of tryptophan is converted into serotonin, and excretion of 5-hydroxy indole acetic acid/(5 HIAA) which is the main catabolite of serotonin is increased in urine.
3- Conversion to melatonin:
It is a hormone, derived from Trp, serotonin is an intermediate. Melatonin is formed in the pineal body tissue and in peripheral nerves of man and secreted into the blood and CSF. Circulating melatonin is taken up by all tissues including the brain to be metabolized and excreted in the urine.
Melatonin is synthesized in human during the dark period of the day and maintained at a low level during the daylight. This diurnal variation is brought about by norepinephrine that stimulates pineal cells to synthesize melatonin. Norepinephrine increases CAMP which increases N. acetyl transferase activity.
III- Metabolic Disorders of Tryptophan Metabolism
- Hartnup disease.
- Vitamin B6 deficiency: increases excretion of xanthurenic acid in urine due to inhibition of kynureninase enzyme.
Nucleotide metabolism
Importance:
Nucleotides (ribonucleoside and deoxyribonucleoside phosphates) are essential for all cells.
- Without these compounds, neither DNA nor RNA can be produced, proteins can’t be synthesized therefore cells can’t proliferate.
- In addition, nucleotides play an extremely important role as “energy currency in the cell.
- Nucleotides serve as carriers of activated intermediates in the synthesis of some carbohydrates, lipids, and proteins and are structural components of a number of essential cofactors such as coenzyme A, FAD, NAD+, and NADP+.
Digestion and Absorption
Most dietary nucleic acids are ingested in the form of nucleoproteins from which the nucleic acids (DNA and RNA) are liberated in the intestinal tract by the action of proteolytic enzymes. Nucleic acids released from ingested nucleoproteins in the intestinal tract are degraded to mononucleotides by ribonucleases, deoxyribonucleases and polynucleotidases.
Nucleotidases and phosphatases hydrolyze the mononucleotides to nucleosides, which either are absorbed or are further degraded by intestinal nucleosidase to purine bases (adenine [A] and guanine [G]), and pyrimidine bases (cytosine [C], thymine [T] and uracil [U]) and pentoses (ribose or 2 dexoyribose).
The dietary purines and pyrimidines are not used to a large extent for the synthesis of tissue nucleic acids but are largely catabolized in intestinal mucosal cells. Also, the pentoses (ribose or 2-deoxyribose) are not utilized by the body due to the absence of specific pentose kinase in the human body.
Nucleotides (or purine and pyrimidine bases found in them) can be synthesized de novo, Also, they can be obtained through salvage pathways that allow the reuse of the preformed bases resulting from normal cell turnover.
You can subscribe to Science Online on YouTube from this link: Science Online
You can download Science online application on Google Play from this link: Science online Apps on Google Play
Function of Calcium, phosphorus, Copper, Zinc and Iodine in human body
Vitamins types & function, Sources of vitamin A & Deficiency of vitamin E
Water soluble vitamins, Vitamin B complex benefits, side effects, sources & deficiency
Benefits of taking Vitamin B complex & vitamin C, sources of Vitamin B6, Biotin & Lipoic Acid
Mineral metabolism, types, disorders, function of Sodium and Potassium